Modern Atomic Theory
|
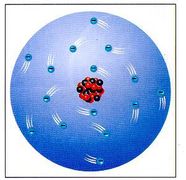
Atoms are composed of:
1) protons - positively charged particles located within the nucleus. 2) neutrons - uncharged particles located within the nucleus. 3) electrons - negatively charged particles which orbit the nucleus. in the picture to the left, the red particles are the protons, the black particles are the neutrons, and the blue particles orbiting are the electrons. |
Ions
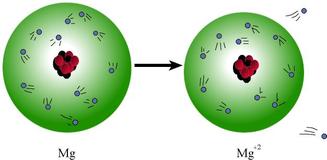
When an atom gains or loses one or more electrons, it acquires an electrical charge. These unbalanced atoms are called ions.
A magnesium atom loses two electrons. Upon losing the two electrons, the atom no longer has a balance of protons and electrons (12 positives and 12 negatives). Now, the atom has 2 more protons than electrons, giving the atom a +2 charge.
When an atom gains or loses one or more electrons, it acquires an electrical charge. These unbalanced atoms are called ions.
A magnesium atom loses two electrons. Upon losing the two electrons, the atom no longer has a balance of protons and electrons (12 positives and 12 negatives). Now, the atom has 2 more protons than electrons, giving the atom a +2 charge.
If it loses electrons, it becomes more positive, and this is called a cation. (positive charge)
On the other hand, if an atom gains extra electrons, thereby having more electrons than protons, it will retain a negative charge. If it gains electrons, it becomes more negative, and this is called an anion. (negative charge)
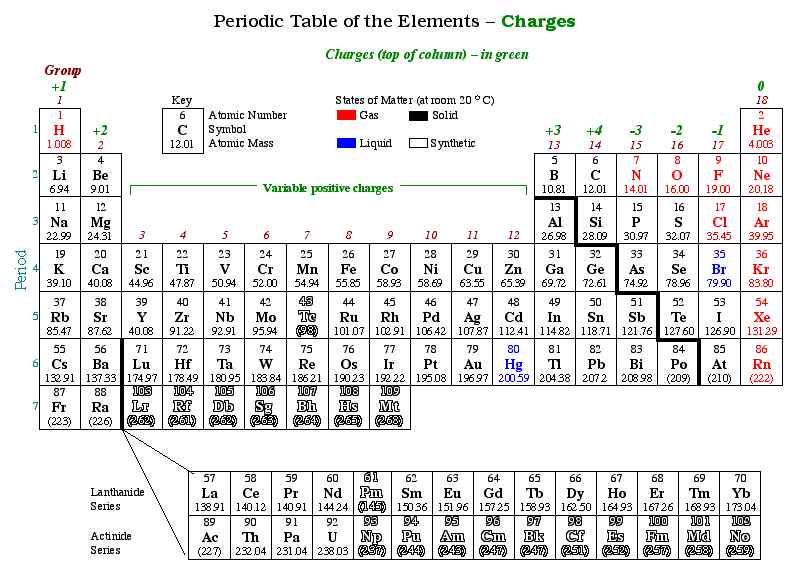
Ionic Charge & the Periodic Table
The periodic table is set-up in such a way that similar atoms are placed in columns. These columns, commonly called families, will have similar chemical properties, acting the same way in the presence of other chemicals. Because of this, we can make generalizations on the charges of the different families.
On the other hand, if an atom gains extra electrons, thereby having more electrons than protons, it will retain a negative charge. If it gains electrons, it becomes more negative, and this is called an anion. (negative charge)
Ionic Charge & the Periodic Table
The periodic table is set-up in such a way that similar atoms are placed in columns. These columns, commonly called families, will have similar chemical properties, acting the same way in the presence of other chemicals. Because of this, we can make generalizations on the charges of the different families.