ionic Compounds
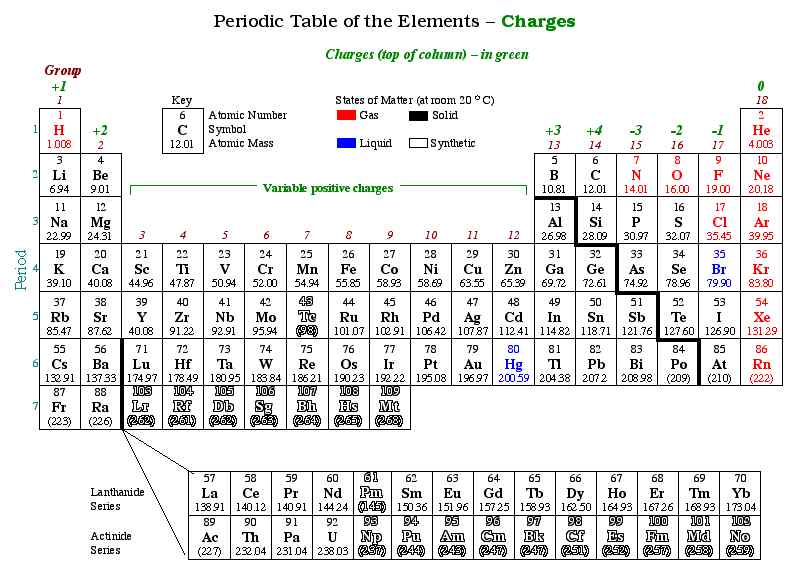
In an ionic bond, a positively charged ion is attracted to a negatively charged ion. Like charges repel, so an ionic bond cannot contain two positively charged ions. Some elements always make specific charges, and some make multiple charges. The periodic table below illustrates this. The green number at the top of the column will help you to determine the charge of the elements in that column:
Ionic compounds are electrically neutral, so there must be a balance of cations and anions in a compound. When combining, positive ions collect enough negative ions to offset their charge. In any compound, the number of atoms of a ion depends on the ion it is bonding with.
Writing Ionic Compound Formulas
There are 4 steps in writing chemical formulas:
1) Determine the ions listed in the name.
2) Write the symbols with the charges above it. (Cation comes first)
3) Determine the number of each ions needed to add up to zero charge.
4) Write the number of each ion as a subscript following the symbol.
Example: sodium sulfide
Step 1: sodium and sulfur are the ions
Step 2: +1 -2
Na S
Step 3: +1 -2
2 Na 1 S --- it is necessary to have two (+1) sodium ions to cancel out the one (-2) sulfur ion.
Writing Ionic Compound Formulas
There are 4 steps in writing chemical formulas:
1) Determine the ions listed in the name.
2) Write the symbols with the charges above it. (Cation comes first)
3) Determine the number of each ions needed to add up to zero charge.
4) Write the number of each ion as a subscript following the symbol.
Example: sodium sulfide
Step 1: sodium and sulfur are the ions
Step 2: +1 -2
Na S
Step 3: +1 -2
2 Na 1 S --- it is necessary to have two (+1) sodium ions to cancel out the one (-2) sulfur ion.
Step 4: Na2S1 or simply Na2S (The 1 is redundant. If you list the symbol, you are assuming you have at least one of that ion.)
Example 2: magnesium nitride
Step 1: magnesium and nitrogen are the ions
Step 2: +2 -3
Mg N
Step 3: +2 -3
3 Mg 2 N --- it is necessary to have three (+2) magnesium ions to cancel out two (-3) nitrogen ions. That way we have +6 and -6
Step 1: magnesium and nitrogen are the ions
Step 2: +2 -3
Mg N
Step 3: +2 -3
3 Mg 2 N --- it is necessary to have three (+2) magnesium ions to cancel out two (-3) nitrogen ions. That way we have +6 and -6
Step 4: Mg3N2
|
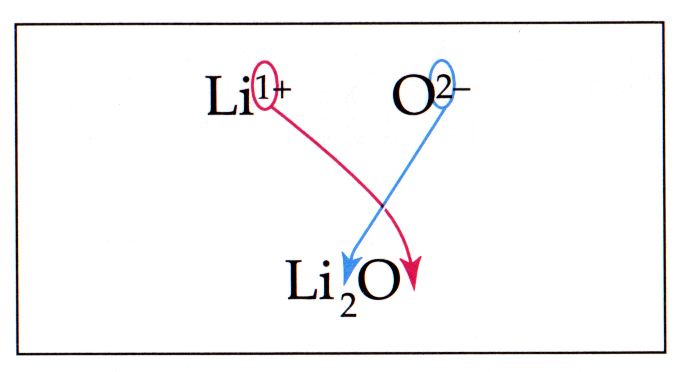
Altenative Method: The Criss-Cross Method In the criss-cross method, the number on the charge for the first ion becomes the number subscript for the second ion. The number on the charge for the second ion becomes the number subscript for the first ion. In the above picture, the number 2 from the charge of the oxygen becomes the subscript for the lithium, and the number 1 from the charge of the lithium becomes the subscript for the oxygen. The criss cross method will work approximately 90% of the time. The conditions when it will not work is when the charges already offset (such as +2 and -2), or when the charges are multiples of each other (such +6 and - 3). In each of these cases the original procedure will reduce the subscripts down. |
Polyatomic Ions
Frequently, atoms can combine to form polyatomic ions. Polyatomi ions are groups of atoms that have an overall charge that needs to be balanced. They act the same as monatomic ions (one atom ions).
Link to List of Common Polyatomic Ions
Example: magnesium nitrate
Step 1: magnesium and nitrate are the ions
Frequently, atoms can combine to form polyatomic ions. Polyatomi ions are groups of atoms that have an overall charge that needs to be balanced. They act the same as monatomic ions (one atom ions).
Link to List of Common Polyatomic Ions
Example: magnesium nitrate
Step 1: magnesium and nitrate are the ions
Step 2: +2 -1
Mg NO3
Step 3: +2 -1
1 Mg 2 NO3 --- it is necessary to have one (+2) magnesium ion to cancel out two (-1) nitrate ions.
Step 4: Mg(NO3)2 - the parenthesis around the NO3 indicate that you need 2 of the NO3 ions. Without the parenthesis, it would look like NO3 2, which appears like you need 1 nitrogen and 32 oxygens.
Mg NO3
Step 3: +2 -1
1 Mg 2 NO3 --- it is necessary to have one (+2) magnesium ion to cancel out two (-1) nitrate ions.
Step 4: Mg(NO3)2 - the parenthesis around the NO3 indicate that you need 2 of the NO3 ions. Without the parenthesis, it would look like NO3 2, which appears like you need 1 nitrogen and 32 oxygens.
Naming Ionic Compounds
There are 3 steps in writing the chemical names of formulas:
1) Determine which elements (or groups) are in your compound.
2) List the name of the cation as it is.
3) Then follow it by the element anion and replace the last syllable with -ide, unless it is a group, which remains the same
Example 1: CaF2
Step 1: calcium and fluorine
Step 2: calcium
Step 3: calcium fluoride (because fluorine is an element, the -ine is changed to -ide)
Step 2: calcium
Step 3: calcium fluoride (because fluorine is an element, the -ine is changed to -ide)
Example 2: BaSO4
Step 1: barium and sulfate
Step 2: barium
Step 3: barium sulfate (no change to ending because sulfate is a polyatomic group)
Multiple Ions
If the cation can form more than one charge, the name of the cation must have a roman numeral following the name which tells the size of the cation charge.
Example 1: Gold (III) Thiocyanate
Step 1: gold and thiocyanate are the ions
Step 2: +3 -1
Au SCN --- gold is in the middle of the periodic table, so it can have multiple charges. In this case, the (III) indicates that the charge on the gold is 3, and since it is listed first, it must be positive, or +3.
Step 3: +3 -1
1 Au 3 SCN --- it is necessary to have one (+3) gold ion to cancel out three (-1) thiocyanate ions.
Step 4: Au(SCN)3 --- SCN is a group, so in order to have more than one, parenthesis are needed.
Example 2: MoCl3
Example 2: MoCl3
Step 1: molybdenum and chlorine are present
Step 2: molybdenum (III) -- the roman numeral is necessary to tell that the charge is +3. We know the charge is +3, because chlorine ions have a charge of -1 each, and this compound has 3 chorine ions, giving the compound a total negative charge of -3. Since compounds need to be neutral, a +3 must balance out the -3, so molybdenum has a +3 charge.
Step 3: molybdenum (III) chloride
Step 2: molybdenum (III) -- the roman numeral is necessary to tell that the charge is +3. We know the charge is +3, because chlorine ions have a charge of -1 each, and this compound has 3 chorine ions, giving the compound a total negative charge of -3. Since compounds need to be neutral, a +3 must balance out the -3, so molybdenum has a +3 charge.
Step 3: molybdenum (III) chloride