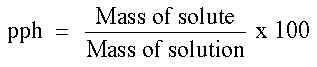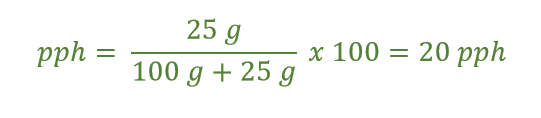Solutions and Concentrations
Solutions
A solution is a homogeneous mixture of two or more substances in a single physical state. There are several properties of solutions:
1) Particles in solution are very small, usually atoms, molecules or ions.
2) Particles are evenly distributed. Every sample of the solution will have the same concentration or ratio of particles as every other sample of the same solution.
3) Particles will not separate out in a solution. Everything stays mixed up evenly.
There are two parts to a solution:
1) Solute - the substance that dissolves. It becomes similar to the solvent. In Kool-Aid, the powder and the sugar are the solutes.
2) Solvent - the substance the solute is dissolving into. In Kool-Aid, the water is the solvent.
There are 4 types of solutions:
1) Solid solutions, also called alloys. Examples are a gold ring, steel, brass.
2) Gas solutions. Example is air
3) Liquid solutions - Examples are gasoline, motor oil.
4) Aqueous solutions, where water is the solvent. These include, orange juice, milk, ocean water.
A solution is a homogeneous mixture of two or more substances in a single physical state. There are several properties of solutions:
1) Particles in solution are very small, usually atoms, molecules or ions.
2) Particles are evenly distributed. Every sample of the solution will have the same concentration or ratio of particles as every other sample of the same solution.
3) Particles will not separate out in a solution. Everything stays mixed up evenly.
There are two parts to a solution:
1) Solute - the substance that dissolves. It becomes similar to the solvent. In Kool-Aid, the powder and the sugar are the solutes.
2) Solvent - the substance the solute is dissolving into. In Kool-Aid, the water is the solvent.
There are 4 types of solutions:
1) Solid solutions, also called alloys. Examples are a gold ring, steel, brass.
2) Gas solutions. Example is air
3) Liquid solutions - Examples are gasoline, motor oil.
4) Aqueous solutions, where water is the solvent. These include, orange juice, milk, ocean water.
Solubility & Saturation
Solubility - The amount of a solute that will dissolve in a specific solvent. A substance may dissolve in a solvent, and it may not. For instance, salt dissolves easily in water: Salt is said to have a high solubility in water. However, sand does not dissolve appreciably in water: Sand has low or no solubility in water. If a substance does dissolve in water, three kinds of solutions can be made:
1) Saturated - A solution is saturated if it contains as much solute as it possibly can. At room temperature, 100 grams of water can dissolve a maximum of 37 grams of salt. When these conditions are achieved, the salt water solution is saturated.
2) Unsaturated - A solution that has less than the maximum amount of solute that can be dissolved. At room temperature, any amount of salt less than 37 grams in 100 grams of water would constitute an unsaturated solution.
3) Supersaturated - A solution that contains more solute than it should, which results in a highly unstable solution. If we were somehow able to get 40 grams of salt dissolved in water at room temperature, the solution would be supersaturated.
How to make a supersaturated solution:
Rock Candy is made from a supersaturated solution of sugar water. Sugar is dissolved in a big kettle of water until no more sugar dissolves at room temperature. Then, the saturated solution is heated up. The higher temperature allows for more sugar to be dissolved, so more sugar is put in. As more and more sugar is put into the kettle, it dissolves, which makes the solution saturated at the new higher temperature. Now, if the sugar is cooled down very slowly, and no more sugar is added, the sugar will stay dissolved. As the temperature cools, the sugar dissolved at that higher temperature stays dissolved, and a supersaturated solution is made. At this lower temperature, the solution is unstable, and by putting a small crystal of solid sugar in the kettle, the extra sugar that is dissolved will attach itself to the sugar, and grow crystals, or rock candy.
Solubility - The amount of a solute that will dissolve in a specific solvent. A substance may dissolve in a solvent, and it may not. For instance, salt dissolves easily in water: Salt is said to have a high solubility in water. However, sand does not dissolve appreciably in water: Sand has low or no solubility in water. If a substance does dissolve in water, three kinds of solutions can be made:
1) Saturated - A solution is saturated if it contains as much solute as it possibly can. At room temperature, 100 grams of water can dissolve a maximum of 37 grams of salt. When these conditions are achieved, the salt water solution is saturated.
2) Unsaturated - A solution that has less than the maximum amount of solute that can be dissolved. At room temperature, any amount of salt less than 37 grams in 100 grams of water would constitute an unsaturated solution.
3) Supersaturated - A solution that contains more solute than it should, which results in a highly unstable solution. If we were somehow able to get 40 grams of salt dissolved in water at room temperature, the solution would be supersaturated.
How to make a supersaturated solution:
Rock Candy is made from a supersaturated solution of sugar water. Sugar is dissolved in a big kettle of water until no more sugar dissolves at room temperature. Then, the saturated solution is heated up. The higher temperature allows for more sugar to be dissolved, so more sugar is put in. As more and more sugar is put into the kettle, it dissolves, which makes the solution saturated at the new higher temperature. Now, if the sugar is cooled down very slowly, and no more sugar is added, the sugar will stay dissolved. As the temperature cools, the sugar dissolved at that higher temperature stays dissolved, and a supersaturated solution is made. At this lower temperature, the solution is unstable, and by putting a small crystal of solid sugar in the kettle, the extra sugar that is dissolved will attach itself to the sugar, and grow crystals, or rock candy.
|
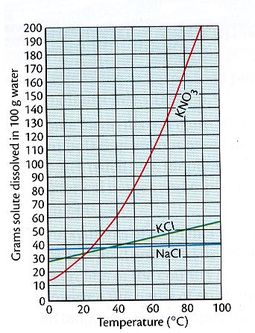
Solubility can be affected by the temperature of the solvent. The graph at right shows the solubility of several solids at different temperatures. For 100 g of water, used as a solvent, any point on the curve is saturated. Therefore, a saturated solution of KNO3 at 25 OC would contain approximately 40 grams of dissolved potassium nitrate. But at 75 OC, more KNO3 will dissolve because of the higher temperature. At 75 OC, 150 g or KNO3 will dissolve in the same amount of water. Any point below the curve indicates an unsaturated solution. At 25 OC, an unsaturated solution of KNO3 would contain less than 40 grams of dissolved potassium nitrate. Any point above the line is a supersaturated solution. At 25 OC, any amount of dissolved KNO3 greater than 40 grams would be supersaturated. |
Concentrations
Chemists often need to specify precisely how concentrated or dilute a solution is. The concentration is the amount of solute dissolved in a given amount of solution. There are numerous ways of expressing concentration:
1) Percent or pph (parts per hundred) - tells the grams of solute per 100 grams of solution
Chemists often need to specify precisely how concentrated or dilute a solution is. The concentration is the amount of solute dissolved in a given amount of solution. There are numerous ways of expressing concentration:
1) Percent or pph (parts per hundred) - tells the grams of solute per 100 grams of solution
For smaller concentrations, these can be used:
2) ppm - parts per million – grams of solute per 1 million grams of solution molecules.
3) ppb - parts per billion - grams of solute per 1 billion grams of solution molecules
In each of these the formula is the same as pph, but instead of multiplying by a hundred, you need to multiply by 1,000,000 and 1,000,000,000 instead, respectively.
Example: A sugar solution is made by dissolving 25.0 g of sugar in 100. g of water. What is the pph of the solution?
Answer: We take the mass of the solute (sugar) and divide it by the mass of the whole solution, which is the mass of the water added to the mass of the sugar.