Properties & Elements
|
Properties of matter
Physical properties are properties of a substance that are observed without altering substance. When you measure the mass or conductivity of a material, you can determine its mass or conductive properties without altering the chemical make up of the substance. After you measure the mass of a sample of sugar, the material will still be sugar. Other examples of physical properties include density, color, melting point, solubility, etc. Physical Changes are a change in the material without changing the material into another substance. We could take frozen water, or ice, and melt it. When you do, the material starts off as the chemical water, and ends up as water. Other examples of physical changes include state changes, crushing, tearing and dissolving. |
|
Chemical properties are properties of a substance observed when altering the substance. Anything observed by changing material A into material B would be a chemical property. For example, when methane gas is burned, the methane is chemically changed into other gases (carbon dioxide and water vapor). A chemical property of methane is that it is flammable. The fact that methane is flammable could not be observed without chemically changing methane into something else. Other examples of chemical properties include cooking and rusting.
Chemical Changes are a change in a material that changes the material into a new substance. When an antacid tablet is placed in water, the tablet reacts with the water and produces bubbles. The bubbles of carbon dioxide formed are due to a reaction of a chemical in the tablet that releases CO2. The new substances formed cannot be changed back into the original materials without another chemical reaction. Other examples of chemical changes would be indicated by many of the following changes: color change, bubbling, fizzing, burning, rusting, change in temperature, etc. |
Conservation of Matter
With all these changes, the law of conservation of matter is always followed. The law of conservation of matter states that matter is neither created nor destroyed in any process.
When 10 g of sugar is placed in 100 g of water, the sugar will dissolve. The sugar will seemingly disappear in the water. We know it is still there for 2 reasons: the mass of the combination is 110 g, and if the water were boiled off, when all the water is evaporated, we will be left with 10 g of sugar.
In a chemical reaction, the law of conservation of matter is obeyed as well. When an antacid tablet is placed in water with a lid on the container, the mass 3.2 g antacid tablet and the 100 g of water will have a total mass of 103.2 g during the entire reaction. If the lid is removed, the mass will go down to 102.8 g. The reason for this 0.4 g difference is the mass of the gas that escapes. When the lid is removed, the gas can enter the atmosphere around the beaker, and the mass of that gas is no longer a part of the system. However, the mass still exits somewhere, just not in the system we are measuring.
With all these changes, the law of conservation of matter is always followed. The law of conservation of matter states that matter is neither created nor destroyed in any process.
When 10 g of sugar is placed in 100 g of water, the sugar will dissolve. The sugar will seemingly disappear in the water. We know it is still there for 2 reasons: the mass of the combination is 110 g, and if the water were boiled off, when all the water is evaporated, we will be left with 10 g of sugar.
In a chemical reaction, the law of conservation of matter is obeyed as well. When an antacid tablet is placed in water with a lid on the container, the mass 3.2 g antacid tablet and the 100 g of water will have a total mass of 103.2 g during the entire reaction. If the lid is removed, the mass will go down to 102.8 g. The reason for this 0.4 g difference is the mass of the gas that escapes. When the lid is removed, the gas can enter the atmosphere around the beaker, and the mass of that gas is no longer a part of the system. However, the mass still exits somewhere, just not in the system we are measuring.
Types of Elements
On the periodic table, there are three types of elements: metals, nonmetals and metalloids. These types are categorized by their properties.
Metals have the following properties:
Nonmetals have the following properties:
Metalloids have properties of both metals and nonmetals. These elements are a buffer between the metals and nonmetals. These elements are defined as having some properties of both metals and nonmetals. A metalloid may be shiny, but not conduct electricity and break when hit with a hammer. Or a metalloid may be malleable and shiny, but not react with acids.
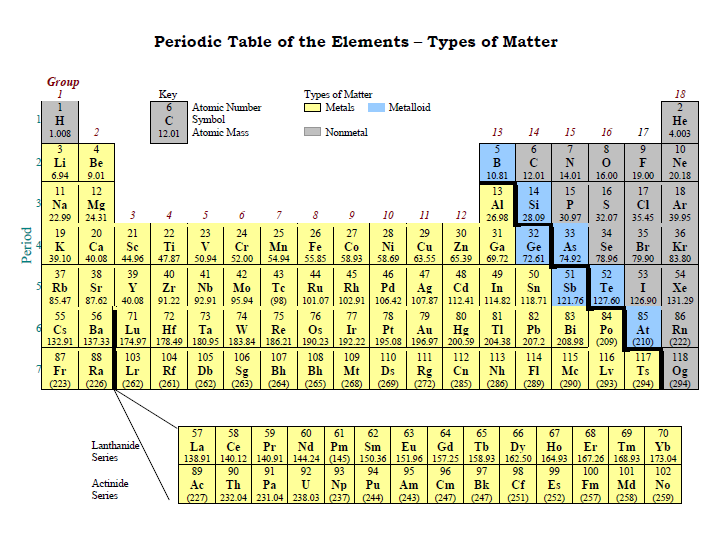
The following periodic table divides the elements of the periodic table into their three types.
On the periodic table, there are three types of elements: metals, nonmetals and metalloids. These types are categorized by their properties.
Metals have the following properties:
- metals have a luster or shine
- metals are good conductors of heat/electricity
- most metals are solids at room temperature (all except mercury)
- metals are malleable, or can be shaped or bent and the metal will not break.
- most metals will react with acids
Nonmetals have the following properties:
- nonmetals do not have a luster; they are dull in appearance
- nonmetals are poor conductors of heat and electricity
- nonmetals are not malleable: when pressure is placed on them, the nonmetals will break.
- nonmetals can be solid, liquid or gas at room temperature
- most nonmetals will not react with acids
Metalloids have properties of both metals and nonmetals. These elements are a buffer between the metals and nonmetals. These elements are defined as having some properties of both metals and nonmetals. A metalloid may be shiny, but not conduct electricity and break when hit with a hammer. Or a metalloid may be malleable and shiny, but not react with acids.
The following periodic table divides the elements of the periodic table into their three types.