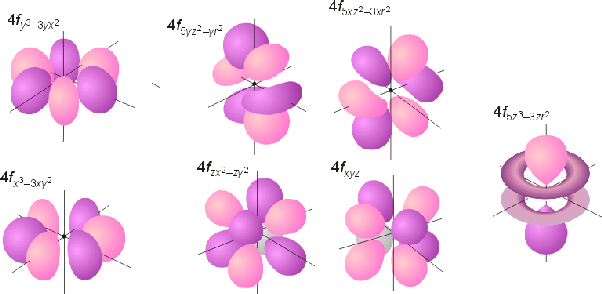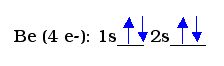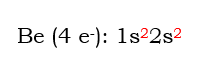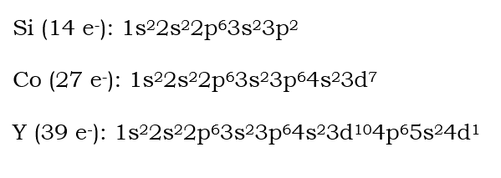Orbitals & Configurations
|
Electron Clouds
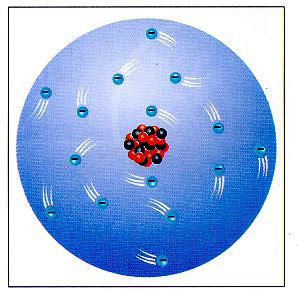
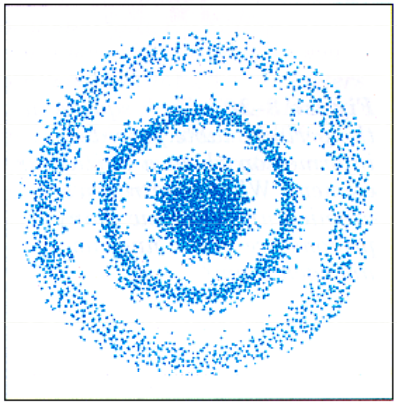
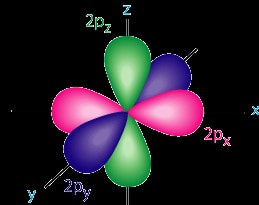
The traditional solar system model of the atom is not completely correct. Although atoms do contain a nucleus with protons and neutrons, and electrons orbit around the nucleus, the idea that electrons have defined orbits is incorrect. It is impossible to identify an electrons position or direction because of the speed of the electron. Electrons travel at speeds approaching the speed of light. Since we cannot identify an electron’s position, we can identify distinct probabilities where an electron can be found in an atom. It is in the form of an electron cloud around the nucleus. In the picture at left, electrons are very likely to be found in the “cloud” of blue dots. Electrons are not likely to be found in the white areas. It is important to remember that an atom is three-dimensional, therefore these circles of high density are actually spheres around the nucleus. Orbitals Within this electron cloud, there are regions in which electrons are found, called an orbital. An orbital is a region around the nucleus where an electron of given energy is found. Orbitals tell the region of where 90% of the electrons are going to be. Orbitals have characteristic shapes and sizes. There are four types of orbitals, designated s, p, d and f. |
d-orbitals
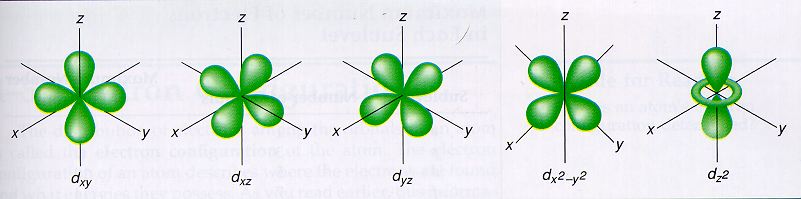
d-orbitals hold up to 10 electrons, with each of the 5 sub-orbital shapes holding 2 electrons each.
d-orbitals hold up to 10 electrons, with each of the 5 sub-orbital shapes holding 2 electrons each.
f-orbitals
f-orbitals hold up to 14 electrons, with each of the 7 sub-orbital shapes holding 2 electrons each.
f-orbitals hold up to 14 electrons, with each of the 7 sub-orbital shapes holding 2 electrons each.
Orbital Diagrams
The distribution of electrons among the orbitals is called the orbital diagram of the atom. These are determined by distributing the electrons in the orbital levels and sublevels based on a set of principles. An orbital diagram shows where each electron can be found within an atom of the element. The electrons are places based on a set of principles:
1) The Aufbau Principle - Electrons are added one at a time to the lowest energy level possible. Translation: Fill the lowest energy level first, then move to next higher. The order in which the orbitals are filled is as follows:
1s 2s 2p 3s 3p 4s 3d 4p 5s 4d 5p 6s 4f 5d 6p 7s 5f 6d 7p
The numbers represent the energy level of the orbit (energy level 1 is closest to the nucleus, 7 is farthest). The letter represent the orbital (the shape within which the electrons orbit).
2) Pauli Exclusion Principle - Each shape within an orbital can hold a maximum of 2 electrons. Electrons within an shape must spin in opposite directions. Electrons must move in opposite directions, one up, one down.
3) Hund’s Rule - Electrons occupy equal energy levels so that a maximum number of unpaired electrons result. Electrons, because they are negative, will repel one another, so if they can be by themselves, they will be. However, they can only be apart until the Aufbau principle takes over. Electrons will fill one in each shape in a p, d, or f orbital, then a second in each shape.
Examples of Orbital Diagrams:
Example 1:
The distribution of electrons among the orbitals is called the orbital diagram of the atom. These are determined by distributing the electrons in the orbital levels and sublevels based on a set of principles. An orbital diagram shows where each electron can be found within an atom of the element. The electrons are places based on a set of principles:
1) The Aufbau Principle - Electrons are added one at a time to the lowest energy level possible. Translation: Fill the lowest energy level first, then move to next higher. The order in which the orbitals are filled is as follows:
1s 2s 2p 3s 3p 4s 3d 4p 5s 4d 5p 6s 4f 5d 6p 7s 5f 6d 7p
The numbers represent the energy level of the orbit (energy level 1 is closest to the nucleus, 7 is farthest). The letter represent the orbital (the shape within which the electrons orbit).
2) Pauli Exclusion Principle - Each shape within an orbital can hold a maximum of 2 electrons. Electrons within an shape must spin in opposite directions. Electrons must move in opposite directions, one up, one down.
3) Hund’s Rule - Electrons occupy equal energy levels so that a maximum number of unpaired electrons result. Electrons, because they are negative, will repel one another, so if they can be by themselves, they will be. However, they can only be apart until the Aufbau principle takes over. Electrons will fill one in each shape in a p, d, or f orbital, then a second in each shape.
Examples of Orbital Diagrams:
Example 1:
In the above orbital diagram, there are several components. 1s stands for the s-shaped orbital in the first energy level. Within that orbital, there are 2 electrons shown by the arrows. The arrows are shown pointing in opposite directions to indicate the opposing spin within the orbital. Since beryllium has 4 electrons, we need to place 4 electrons. Only 2 electrons can fit in the 1s orbital, so we move up to the next orbital, and place the next 2 electrons in the 2s.
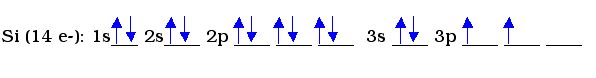
Example 2:
Example 2:
ilicon requires more orbitals. It is then necessary to move farther up the sequence listed for the Aufbau principle. The 2p orbital has three blanks because of the three different shapes within a p-orbital. Each blank can hold up to 2 electrons. In the 3p orbital, only 2 electrons need to be placed. Those 2 electrons are not in the same blank because they repel one another and would prefer to be apart.
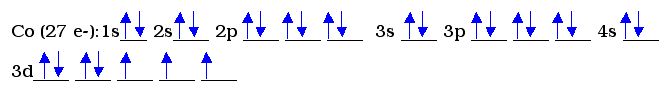
Example 3:
Example 3:
Cobalt requires still more orbitals. The 3d orbital has 5 blanks for the 5 shapes possible within a d-orbital. The d-orbital fills in one in each of the 5 shapes, and then starts doubling up electrons.
Electron Configurations
An electron configuration is a shortened version of an orbital diagram. An electron configuration tells the reader how many electrons are placed in each orbital. An example of an electron configuration is as follows:
Electron Configurations
An electron configuration is a shortened version of an orbital diagram. An electron configuration tells the reader how many electrons are placed in each orbital. An example of an electron configuration is as follows:
The configuration above simply states that there are 2 electrons in the 1s orbital and 2 electrons in the 2s orbital. The exponents tell the number of electrons there are in the orbital. It can be compared to the Example 1 in the Orbital Diagram section. The configuration is a simplified version of the orbital diagram. It assumes you understand that the different orbitals have different shapes and where they are located.
More examples:
More examples: