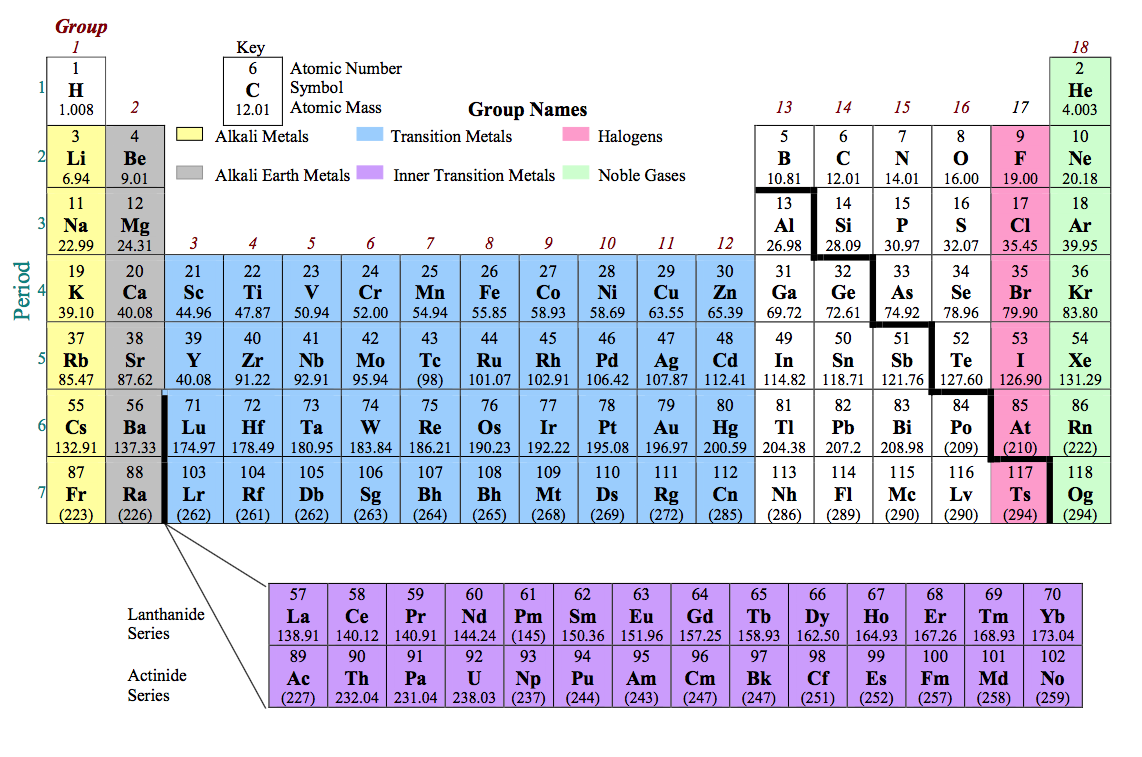
Special Group and Period Names
Many groups and periods on the periodic table have been give special names. Although it may be common to refer to the first column of the periodic table (Li, Na, K, etc.) by “Group 1”, the group has a special name. On the periodic table below, the groups are numbered to help identify the element groups and their names:
|
Group 1 (Group 1A) - Alkali Metals Elements of this group are shown at left. The elements are highly reactive due to the single s-orbital electron. Each element in this group forms a +1 charge. These elements react with oxygen & water rapidly, and must be stored in oil in element form. Alkali metals are soft metals, and very abundant in compound form. Group 2 (Group 2A) - Alkali Earth Metals Elements of this group are shown at left. The elements are very reactive, but not as much as the alkali metals. Each element in this group forms a +2 charge. Alkali earth metals are soft metals, and very abundant in compound form |
|
Group 17 (Group 7A) - Halogens
Elements of this group are shown at left. The elements are also highly reactive, this time due to the 5 p-orbital electrons. These elements are very electronegative, and attract electrons very easily. Each element in this group forms a -1 charge. These nonmetals are very common in compound form, and can be a gas, liquid or solid at room temperature. Group 18 (Group 8A) - Noble Gases Elements of this group are shown at right. These elements are gases with a full p-orbital. Very few compounds of noble gases made due to the stable p-orbital. These elements are commonly used in gas tubes for neon-type signs. |
Groups 3 - 12 - Transition Metals (d-orbital)
Elements of these type are shown below. These elements are metals found throughout the Earth, sometimes in elemental form. The charges of these elements vary, but most commonly form a +2 charge. Most applications of industry come from these elements.
Elements of these type are shown below. These elements are metals found throughout the Earth, sometimes in elemental form. The charges of these elements vary, but most commonly form a +2 charge. Most applications of industry come from these elements.
Group 3 - Inner Transition Metals: (f-orbital)
Elements of these type are shown below. This group of highly unstable elements are largely used in nuclear reactions and lighting. The elements are unstable due to the size of the atoms.
Elements of these type are shown below. This group of highly unstable elements are largely used in nuclear reactions and lighting. The elements are unstable due to the size of the atoms.