Periodic Trends
The periodic table and its setup has told us many things, and we can make generalizations about atoms and their properties based on its setup. These generalizations are called periodic trends. These trends can be predicted based on the periods and groups the elements are in. Groups are the vertical columns on the periodic table. Periods are the horizontal rows on the periodic table.
Atomic Radius (Atom Size)
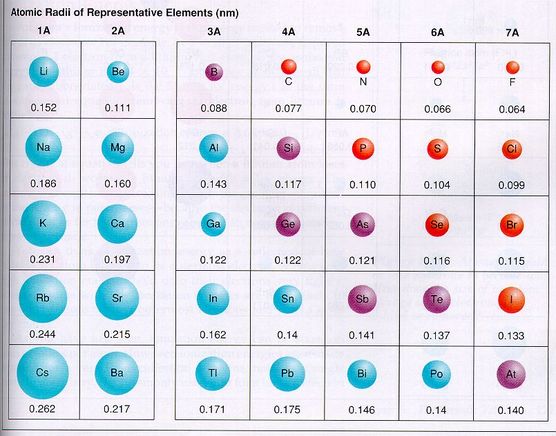
Atomic radius - the distance from the center of the atom to the outer edge. The data for the s and p orbitals are pictured in the picture below:
Atomic radius - the distance from the center of the atom to the outer edge. The data for the s and p orbitals are pictured in the picture below:
From this diagram, we see that as you move down a group, the radius of the atom becomes larger. This should make sense, because the elements contain more protons, neutrons and electrons as you go down the column. Also, the number of orbits (energy levels and orbitals) increases as you go down a group. Lithium has a configuration of 1s22s1, so its farthest out electron is in the second energy level, or orbit. Sodium has a configuration of 1s22s22p63s1, so its farthest out electron is in the third energy level, making it one orbit bigger.
From the diagram, we see that as you move across the period, from left to right, the atoms get smaller. This goes against are thinking that as you increase the electrons, the atom should get bigger. The reason for this is that as you move left to right across the period, there are more protons in the nucleus, which give a stronger pull on the outermost electrons. The protons increase, but so do the electrons. But the electrons, say for chlorine, are still in the 3rd energy level, the same as sodium. Since the orbit stays the same, but the protons pull harder on the orbit, the orbit will then get sucked in closer to the nucleus, due to the stronger attraction between more protons and electrons. This stronger attraction pulls the electron orbits in closer to the nucleus, making the atom smaller.
In summary, the trends for atomic radius are:
Atomic radius increases down a group.
Atoms get smaller as you move across the period.
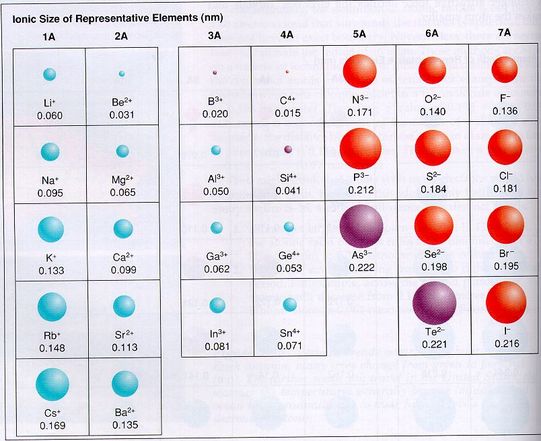
Ionic Radius (Size of Ions)
Ionic Radius - the radius of an element in its most common charged form.
In summary, the trends for atomic radius are:
Atomic radius increases down a group.
Atoms get smaller as you move across the period.
Ionic Radius (Size of Ions)
Ionic Radius - the radius of an element in its most common charged form.
Again, looking at the s and p orbitals, we find that the atom size changes slightly when the neutral atom becomes charged. As we know, group 1A elements form a +1 charge. Compared to the neutral atomic radius from above, we find the atom has shrunk. The reason for this is 2 fold:
1) The atom has lost part of it’s mass to acquire a positive charge. The atom has lost an electron to become positive, therefore it should be smaller.
2) Now that the atom has less electrons, the protons in the center of the atom can pull harder on the reduced number of electrons, and shrink the atom.
As the charge becomes more positive, losing more and more electrons, the radius get smaller and smaller, due to the same reasons as above.
On the other hand, when group 5A gains a -3 charge, we notice that the atom becomes bigger in radius. Again the reason for this is two-fold.
1) The atom has gained more mass to acquire a positive charge. The atom has gained electrons to become negative, therefore it should be bigger.
2) Now that the atom has more electrons, the protons in the center of the atom have a weaker pull on the increased number of electrons, and the atom grows larger.
In summary, the trends of atomic radius are:
When atoms lose electrons, their radius decreases.
When atoms gain electrons, their radius increases.
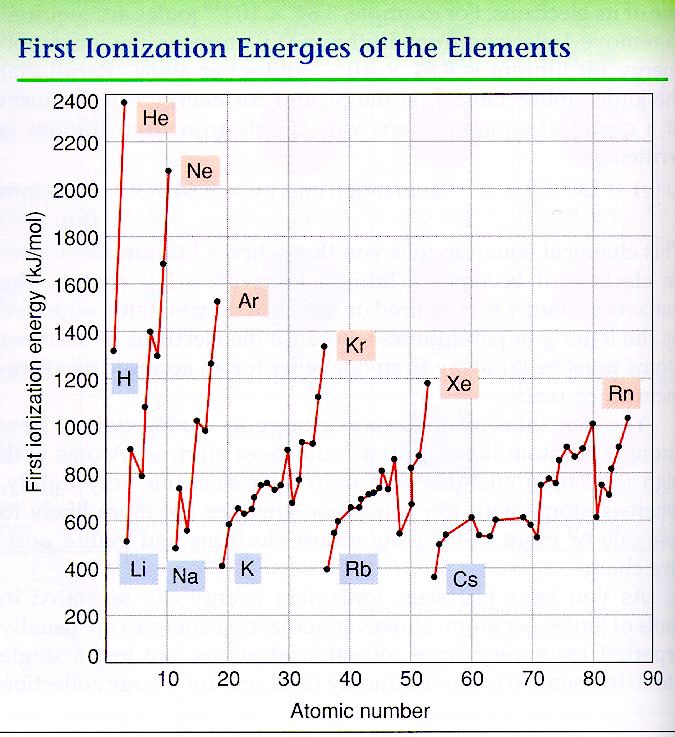
First Ionization Energy
Ionization energy is the energy required to remove one of an atom’s electrons. Look at the graph below.
1) The atom has lost part of it’s mass to acquire a positive charge. The atom has lost an electron to become positive, therefore it should be smaller.
2) Now that the atom has less electrons, the protons in the center of the atom can pull harder on the reduced number of electrons, and shrink the atom.
As the charge becomes more positive, losing more and more electrons, the radius get smaller and smaller, due to the same reasons as above.
On the other hand, when group 5A gains a -3 charge, we notice that the atom becomes bigger in radius. Again the reason for this is two-fold.
1) The atom has gained more mass to acquire a positive charge. The atom has gained electrons to become negative, therefore it should be bigger.
2) Now that the atom has more electrons, the protons in the center of the atom have a weaker pull on the increased number of electrons, and the atom grows larger.
In summary, the trends of atomic radius are:
When atoms lose electrons, their radius decreases.
When atoms gain electrons, their radius increases.
First Ionization Energy
Ionization energy is the energy required to remove one of an atom’s electrons. Look at the graph below.
|
We find that in each period, the elements with the highest ionization energy are group 18 elements, also called the noble gases, all the way to the right on the periodic table. These elements, the noble gases, are called noble because they do not react with any other elements. These elements, due to their full p-orbital, are already stable and do not need to bond to become stable. In a sense, they are the cool kids.
All other elements on the periodic table do not have a full p-orbital, and attempt to get a full p-orbital by bonding with other atoms. The elements gain or lose electrons to get to the same electron configuration as a noble gas, or stable. In a sense, they are trying to be like the cool kids. In order to do this, some atoms will lose electrons. For instance, sodium has 11 electrons. Neon is the closest noble gas with 10 electrons. If sodium were to lose one electron, it would be similar in configuration to neon, a noble gas. Losing one electron is easier than gaining seven electrons to get to 18, the number of electrons as argon. Other atoms may gain electrons, because it would be easier to get to a noble gas electron configuration. In the case of the group 1 elements (Li, Na, K, Rb, etc.), it would not take much energy to pull off an electron because these atoms want to lose an electron to become a stable configuration like a noble gas (or to become cool). Group 17 elements (F, Cl, Br, etc.), on the other hand, want to gain one electron to be like a noble gas. Because of this it would be very difficult, or take a lot of energy, to remove an electron because it really wants an additional electron. |
In summary, the trends of first ionization energy are:
Ionization energies decrease as you move down a group. Ionization energies increase as you move left to right across the period. |
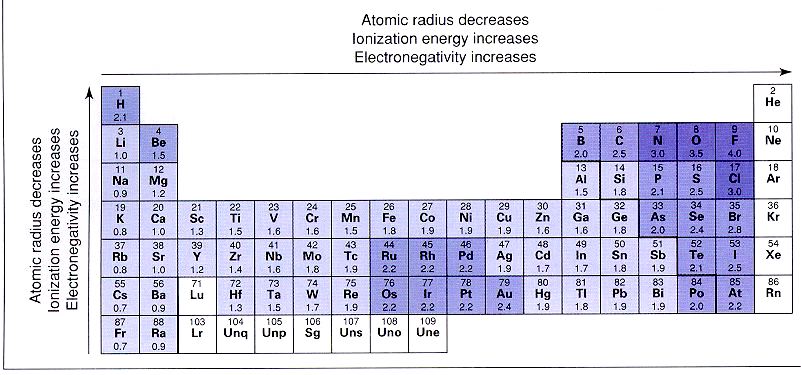
Electronegativity
Electronegativity is an atoms ability to pull electrons toward it. The scale starts at 4.0 for the highest electronegativity and goes down from there.
Electronegativity is an atoms ability to pull electrons toward it. The scale starts at 4.0 for the highest electronegativity and goes down from there.
The electronegativities of noble gases are zero. This should make sense because the noble gases are already stable, and have no desire to pull an electron toward them. The group 1 elements have low electronegativities because they do not want to gain another electron. In fact, they want to get rid of the electron that they have. The highest electronegativities are in the group 17 elements. These elements need just one electron to be like a noble gas, so they really pull hard to get that electron.
In summary, the electronegativity trends are:
Electronegativity decreases as you move down a group.
Electronegativity increases as you move left to right across the period (but the noble gases at the right are zero).
In summary, the electronegativity trends are:
Electronegativity decreases as you move down a group.
Electronegativity increases as you move left to right across the period (but the noble gases at the right are zero).