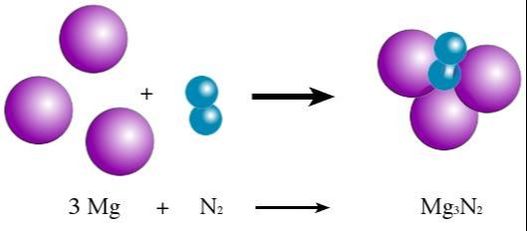Types Of reactions
In this chemistry course, we will focus on five basic types of reactions:
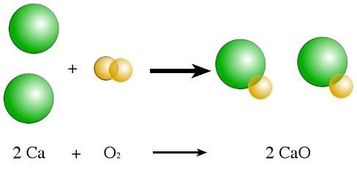
1) Synthesis - Two or more reactants come together to form one product. The reactants are often elements coming together to form a compound, but could also be two (or more) compounds coming together to form 1 larger compound.
1) Synthesis - Two or more reactants come together to form one product. The reactants are often elements coming together to form a compound, but could also be two (or more) compounds coming together to form 1 larger compound.
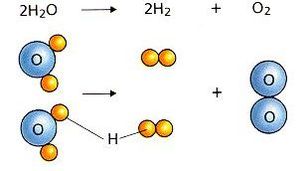
2) Decomposition - A single compound is broken down into two or more smaller compounds or elements.
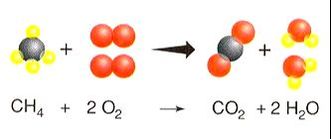
3) Combustion - Reactions in which a hydrocarbon is combined with O2 and burned to always form carbon dioxide and water. A hydrocarbon must contain hydrogen and carbon. The products are always CO2 and H2O.
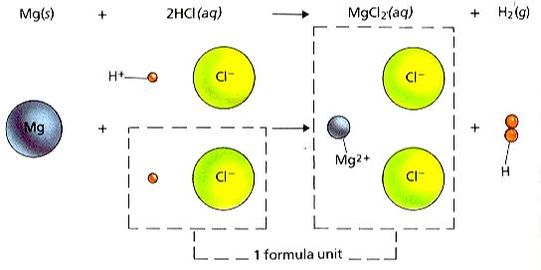
4) Single Replacement - An uncombined element displaces an element that is part of a compound. In ionic compounds, a compound is comprised of a positive ion and a negative ion. In other words, the uncombined element replaces an element of similar charge (a positive ion replaces the positive, a negative ion replaces the negative ion)
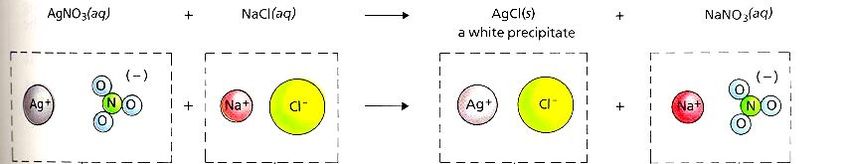
5) Double Replacement - Ions from two different compounds replace each other. Commonly know as the double switch, the positive ions switch the negative ions they are combined with.