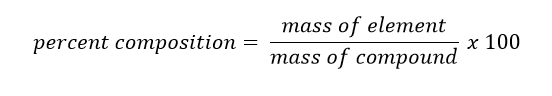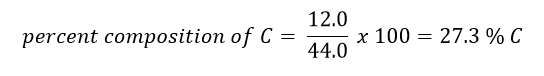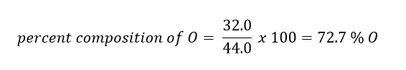Atomic Mass & Molar Mass
Atomic & Formula Mass
As you may recall, protons and neutrons are roughly the same mass, and their mass was given a special unit, the amu. Protons and neutrons were approximately 1 amu each, while an electron was so small it was held to be 0 amu. Using this information, we can conclude that the atomic mass of an element is equal to the atomic mass number in amu.
In other words, the mass of one carbon atom is 12.0 amu, based upon it’s mass on the periodic table. We round the units to the nearest tenths place for all atoms. Copper, therefore would have a mass of 63.5 amu.
As you may recall, protons and neutrons are roughly the same mass, and their mass was given a special unit, the amu. Protons and neutrons were approximately 1 amu each, while an electron was so small it was held to be 0 amu. Using this information, we can conclude that the atomic mass of an element is equal to the atomic mass number in amu.
In other words, the mass of one carbon atom is 12.0 amu, based upon it’s mass on the periodic table. We round the units to the nearest tenths place for all atoms. Copper, therefore would have a mass of 63.5 amu.
Formula mass is a little different. Formula mass is the sum of the atomic masses in a compound. For example, water, H2O has a formula mass of 18 amu.
Reason: A formula of H2O has 2 hydrogens and 1 oxygen. The mass of a hydrogen is 1.0 amu. Since we have two, we multiply 2 x 1.0 amu = 2 amu. The oxygen has a mass of 16.0 amu, according to the periodic table. The total mass is the mass of hydrogen plus oxygen, or 2.0 amu + 16.0 amu = 18.0 amu.
Another example: K3PO4 would be K = (3 x 39.1 amu) + P (1 x 31.0 amu) + O (4 x 16.0 amu) = 212.3 amu
Reason: A formula of H2O has 2 hydrogens and 1 oxygen. The mass of a hydrogen is 1.0 amu. Since we have two, we multiply 2 x 1.0 amu = 2 amu. The oxygen has a mass of 16.0 amu, according to the periodic table. The total mass is the mass of hydrogen plus oxygen, or 2.0 amu + 16.0 amu = 18.0 amu.
Another example: K3PO4 would be K = (3 x 39.1 amu) + P (1 x 31.0 amu) + O (4 x 16.0 amu) = 212.3 amu
Molar Mass
We know how to find the formula and atomic masses. Molar mass is the next step. The molar mass of a compound is the mass of one mole of a compound. The unit for this is g/mol. It is directly related to atomic and formula masses. Whatever the formula mass is in amu, the molar mass is the same number, with the unit g/mol instead.
Examples:
We know how to find the formula and atomic masses. Molar mass is the next step. The molar mass of a compound is the mass of one mole of a compound. The unit for this is g/mol. It is directly related to atomic and formula masses. Whatever the formula mass is in amu, the molar mass is the same number, with the unit g/mol instead.
Examples:
|
Formula
Au NaOH CuCl2 Ca(NO3)2 |
Formula Mass
197.0 amu 40.0 amu 134.5 amu 164.1 amu |
Molar Mass
197 g/mol 40.0 g/mol 134.5 g/mol 164.1 g/mol |
Percent Composition
It is often necessary to know how much of a particular element is in a compound. The percent composition of the mass made up by each element in a compound is called the percent composition of that element. Percent composition can be calculated using the following formula:
It is often necessary to know how much of a particular element is in a compound. The percent composition of the mass made up by each element in a compound is called the percent composition of that element. Percent composition can be calculated using the following formula:
Example: Suppose you had a mole of CO2. What is the percent composition of each element?
Answer: In one mole of CO2, there is 12.0 g of C (due to the one carbon in the compound) and 32.0 g of O (due to two oxygens in the compound - 2 x 16.0 g = 32.0 g). The total mass of one mole of CO2 is therefore 44.0 g (12.0 g C + 32.0 g O).
Answer: In one mole of CO2, there is 12.0 g of C (due to the one carbon in the compound) and 32.0 g of O (due to two oxygens in the compound - 2 x 16.0 g = 32.0 g). The total mass of one mole of CO2 is therefore 44.0 g (12.0 g C + 32.0 g O).