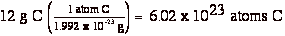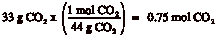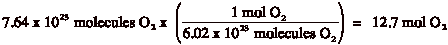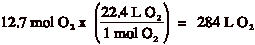The Mole
The mole is a unit of measure, just like a dozen, a gross or a ream. We all know that the word “dozen” means 12 units of something. A dozen eggs equals 12 eggs. A gross is 12 dozen of something, or 144 units. A ream is 500 units. These are all words that mean numbers. A mole is a word that means a number (a very large number).
Mole Determination
To use the atom as the standard unit of measure for the mass is difficult. It is impossible to directly measure the mass of an atom. If we put a single atom on one of science’s most accurate scales, the mass would still read zero. No balance in existence can measure the mass of even the largest atom. An atom does not weigh enough to be measured. So, we would like to use a different measure, the gram.
It would make sense to have the mass of a certain number of atoms the same as the atomic mass number, only in grams instead of amu. In other words, it seems unnecessary to learn yet another number ot associate with carbon. We already know that the atomic number of carbon is 6 and the atomic mass is 12 amu. One atom of carbon weighs 12 amu. We would like the standard for carbon to be 12 g instead of 12 amu. This way we can keep he same number for the mass of carbon, just use a different unit. However, in order to do this, we need to use more than one atom of carbon --- many more!
Mole Determination
To use the atom as the standard unit of measure for the mass is difficult. It is impossible to directly measure the mass of an atom. If we put a single atom on one of science’s most accurate scales, the mass would still read zero. No balance in existence can measure the mass of even the largest atom. An atom does not weigh enough to be measured. So, we would like to use a different measure, the gram.
It would make sense to have the mass of a certain number of atoms the same as the atomic mass number, only in grams instead of amu. In other words, it seems unnecessary to learn yet another number ot associate with carbon. We already know that the atomic number of carbon is 6 and the atomic mass is 12 amu. One atom of carbon weighs 12 amu. We would like the standard for carbon to be 12 g instead of 12 amu. This way we can keep he same number for the mass of carbon, just use a different unit. However, in order to do this, we need to use more than one atom of carbon --- many more!
Since 1 amu = 1.66 x 10-24 g, it is possible through a simple calculation to determine the number of atoms of carbon needed to put on a scale so it will read 12 grams.
1 carbon atom = 12 amu = 12 x 1.66 x 10-24 g = 1.992 x 10-23 g
So, if you put 6.02 x 1023 atoms of C on the scale, it will read 12 g.
This simple calculation, first conducted by Amadeo Avogadro, in the early 1800’s determined that 6.02 x 1023 particles always is the number of particles necessary to get the scale to read the amu number in grams. It is termed Avogadro’s number (n), and will always be the same regardless of what substance you are using.
This number is called the mole. In every mole of a substance, there are 6.02 x 1023 particles.
This number is called the mole. In every mole of a substance, there are 6.02 x 1023 particles.
Examples:
|
Amount
1 atom Au 6.02 x 1023 atoms Au 1 mole Au 2 moles Au |
Mass
197.0 amu 197.0 g 197.0 g 394.0 g |
Amount
1 molecule NaOH 6.02 x 1023 molecules NaOH 1 mole NaOH 2 moles NaOH |
Mass
40.0 amu 40.0 g 40.0 g 80.0 g |
|
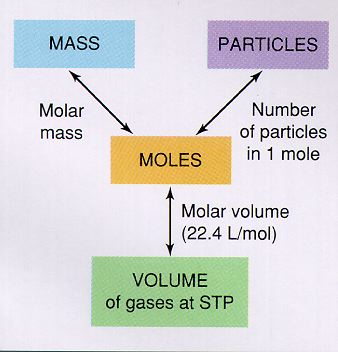
Mole Conversions
Throughout the rest of the year, we will need to know how many moles are in a certain sample of a compound. We may know the mass, moles, volume of gas (at STP) or the # of particles. Using the conversions below, we can then convert the given unit into moles to do further calculations. Prior to that, we need to get comfortable changing units in and out of moles. The diagram at right is very useful in this respect. You will notice that the unit moles is in the center of the diagram. Every unit can be turned into moles, and back out of moles. However, you cannot go from one of the outside units to another without going through moles. Conversion factors
1 mole = 6.02 x 1023 molecules 1 mole = 22.4 L of gas 1 mole = molar mass (mass differs with each element or compound) |
Example: How many moles are present in 33 g of CO2?
Example 2: How many liters of O2 are present in 7.64 x 1024 molecules of O2?
Answer: This is a 2 step process. It is difficult to go directly from molecules to liters. It is necessary to go through moles. Therefore, molecules must be converted to moles first:
Then, you can convert moles into liters: