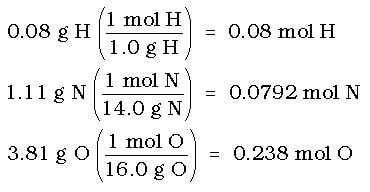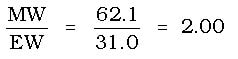Empirical & Molecular Formula
Empirical vs. Molecular Formula
An empirical formula is a formula that gives the simplest whole-number ratio of the atoms of the elements in a compound. An example of an empirical formula would H2O and NaCl. For NaCl, there is one sodium atom for every chlorine atom. In water, there is 2 hydrogen atoms for every oxygen atom, or the ratio of hydrogen to oxygen atoms is 2:1.
The molecular formula is always a multiple of the empirical formula, and how the substance exists. For instance, hydrogen peroxide exists as H2O2. However, H2O2 can be reduced to HO, which is the empirical formula. Hydrogen peroxide only exists as H2O2, never as HO. In fact, HO doesn't even exist as a stable compound.
The molecular formula is always a multiple of the empirical formula, and how the substance exists. For instance, hydrogen peroxide exists as H2O2. However, H2O2 can be reduced to HO, which is the empirical formula. Hydrogen peroxide only exists as H2O2, never as HO. In fact, HO doesn't even exist as a stable compound.
The molecular formula is always a multiple of the empirical formula, and how the substance exists.
Examples of empirical and molecular formulas:
|
Empirical Formulas (simplest)
NaOH
CH2O Na2CO3 NO2 K2CrO4 |
Molecular Formulas (how they exist)
NaOH
C11H22O11 Na2CO3 N2O4 K2CrO4 |
Notice how for sugar, C11H22O11, the molecular formula is 11 times bigger than the empirical formula. The molecular formula is always a multiple of the empirical formula. However, the empirical formula is not always the same compound as the molecular formula. The C11H22O11 is sugar, which is essential for life. CH2O is formaldehyde, which is a toxic substance used in preserving dead bodies for display at a funeral.
Also, notice how sometimes the molecular formula is the same as the empirical formula. In NaOH, the simplest formula is also how the substance exists. The molecular formula is the empirical formula times 1.
Also, notice how sometimes the molecular formula is the same as the empirical formula. In NaOH, the simplest formula is also how the substance exists. The molecular formula is the empirical formula times 1.
Calculating Empirical Formula
By use of the fact that molecule ratios and mole ratios are going to be equal, determining the empirical formula of a substance can be accomplished by calculating a mole ratio of all atoms in the compound. The information for this can be collected in a lab by determining the percent composition of a compound or through mass analysis of the compound.
A simple process to determine the empirical formula would be to follow the steps below:
1) Determine the moles of each element in the compound.
2) Set-up a mole ratio for the elements.
3) Divide each mole number in the ration by the smallest mole value.
4) Round numbers off to nearest whole number, and write the formula.
The process is shown below for the following example:
Example 1: A 5.00 g solid acid sample is analyzed to be 0.08 g hydrogen, 1.11 g nitrogen and 3.81 g oxygen. What is the empirical formula of the compound?
Step 1: Change grams to moles for all substances:
By use of the fact that molecule ratios and mole ratios are going to be equal, determining the empirical formula of a substance can be accomplished by calculating a mole ratio of all atoms in the compound. The information for this can be collected in a lab by determining the percent composition of a compound or through mass analysis of the compound.
A simple process to determine the empirical formula would be to follow the steps below:
1) Determine the moles of each element in the compound.
2) Set-up a mole ratio for the elements.
3) Divide each mole number in the ration by the smallest mole value.
4) Round numbers off to nearest whole number, and write the formula.
The process is shown below for the following example:
Example 1: A 5.00 g solid acid sample is analyzed to be 0.08 g hydrogen, 1.11 g nitrogen and 3.81 g oxygen. What is the empirical formula of the compound?
Step 1: Change grams to moles for all substances:
Step 2: Set up a mole ratio for the elements
H : N : O
0.08 : 0.0792 : 0.238
0.08 : 0.0792 : 0.238
Step 3: Divide each by the smallest - the smallest number in this example is 0.0792, so when you divide each by 0.0792,
H : N : O
0.08 : 0.0792 : 0.238
0.0792
0.08 : 0.0792 : 0.238
0.0792
it changes to
H : N : O
1.01 : 1 : 3.01
1.01 : 1 : 3.01
Step 4: Round off each number to nearest whole number. The numbers in the ratio are the subscripts of the formula:
HNO3
**Note: In most cases, the ratios come out to be on or very near to whole numbers, However, the ratio could be 1:1.5, and this number of 1.53 is a little large to round down to 1 or too small to round up to 2. However, the ratio could be multiplied by 2 to change 1:1.5 to 2:3, which is a simplest whole number ratio.
Calculating Molecular Formulas
In many cases, especially in organic molecules, the empirical formula is not how the compound appears. For instance, butane has the formula C4H10, in which each of the subscripts can be divided by two to be C2H5. Although C2H5 is the empirical formula, butane never appears as this simplest formula, but rather as a multiple of that formula. The formula that gives the actual number of atoms of each element in a compound is called the molecular formula.
In order to find the molecular formula, it is necessary to know two things: the empirical formula of the compound and the molar mass of the compound. Since we know the molecular formula will have the same ratio as the empirical formula, just multiplied by a whole number factor, we can follow these steps to determine the molecular formula:
1) Determine the empirical formula mass.
2) Divide the molar mass of the molecular formula by the empirical formula mass. The number you get is how many times more atoms are in the molecular formula than the empirical.
3) Multiply the subscript of the empirical formula by the factor from step 2 to get the molecular formula.
1) Determine the empirical formula mass.
2) Divide the molar mass of the molecular formula by the empirical formula mass. The number you get is how many times more atoms are in the molecular formula than the empirical.
3) Multiply the subscript of the empirical formula by the factor from step 2 to get the molecular formula.
Example 2: The molar mass of a compound is determined to be 62.1 g/mol, while the empirical formula is CH3O. What is the molecular formula for the compound.
Step 1: The empirical formula mass would be 12.0 + 3(1.0) + 16.0 = 31.0 g/mol
Step 3: Multiply 2(CH3O) = C2H6O2