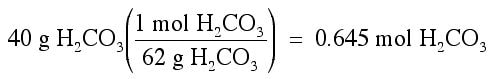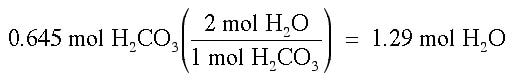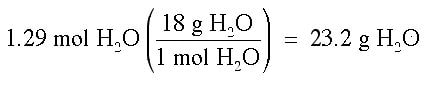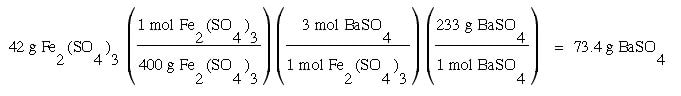Mass-Mass Stoichiometry
Stoichiometry
Mole to mole stoichiometry (as learned in the previous section) is an important component in chemistry, It will allow us to convert from one substance to another within the confines of a chemical reaction. So, lets imagine that we are performing this reaction:
Mole to mole stoichiometry (as learned in the previous section) is an important component in chemistry, It will allow us to convert from one substance to another within the confines of a chemical reaction. So, lets imagine that we are performing this reaction:
H2CO3 + 2 KOH --> K2CO3 + 2 H2O
If we know the moles of one of the substances, we can find the moles of any of the other materials within the equation. However, we usually do measure materials out in moles during a lab. Instead, we typically use mass, or grams. Unfortunately there is not a quick and easy process to go from grams of one material to grams of another material within a balanced chemical reaction. However, there is a way.
Using the above equation, suppose we wanted to know how many grams of water (H2O) can be made from 40 g of H2CO3. We know how to convert moles of H2CO3 into moles of H2O, so we need to convert the 40 grams of H2CO3 into moles of H2CO3. The work looks like this:
Then, we can convert moles of H2CO3 into moles of H2O:
Finally, we do not want our answer in moles, but in grams of H2O, so we complete one more conversion:
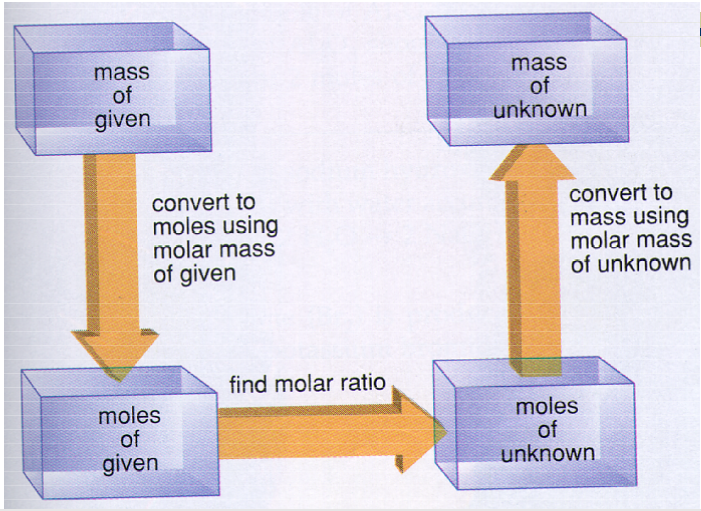
Mass to Mass Stoichiometry requires three steps to complete. A diagram is below to make this process more clear
Another example: How many grams of BaSO4 can be made when 42 g of Fe24(SO4)4 is used in the following balanced chemical equation:
Fe2(SO4)3 + 3 BaS ---> 3 BaSO4 + Fe2S3
Answer: