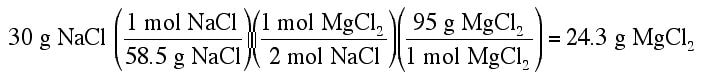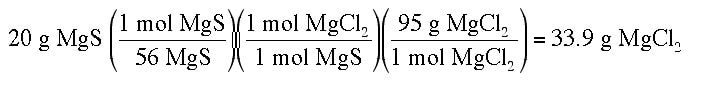Limiting Reactant
|
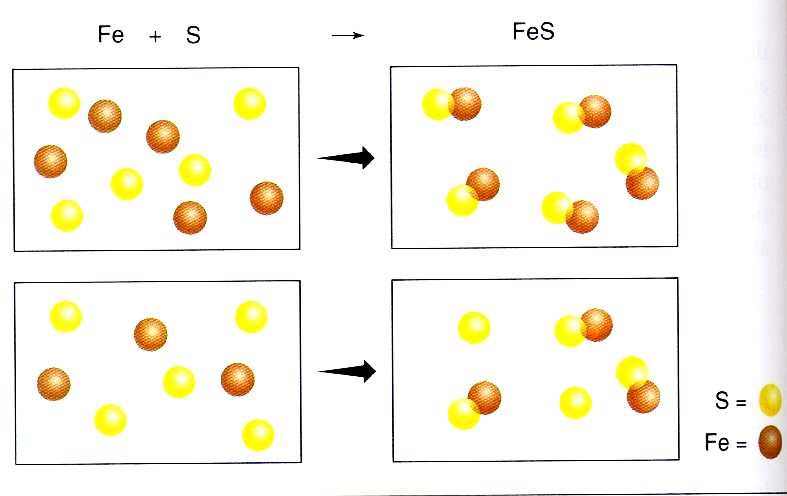
In most chemical reactions, chemicals are combined in non-stoichiometric proportions. This means that you may add more than you need of one reactant than the other. For example, look at the diagram at right. In the picture, iron and sulfur are being mixed. On the left of the arrow they have yet to combine, and on the right of the arrow the elements have combined to form FeS.
In the first reaction, there are 5 iron and 5 sulfur, which is perfect proportions (1:1). Therefore, 5 compounds are made, and there is no iron and sulfur left over. In the second reaction, there are 3 iron and 5 sulfur. When the elements combine, only 3 compounds can be made because there is a shortage of iron. There are two extra sulfur floating around because they have nothing to combine with. In this case, the iron limits the amount of product that can be formed. When this happens, one reactant is completely used up, but there is a excess of the other reactant. The reactant that limits the amount of product that can be formed is called the limiting reactant. |
Let's look at it another way: Suppose you were building bicycles. Each bicycle requires 2 wheels, a frame and a handlebar. If your warehouse inventory contained 324 wheels, 175 frames and 210 handlebars, how many bicycles could be made?
The answer is 162. Since each bicycle requires 2 wheels, you only have enough wheels to make 162 bicycles (324/2 = 162). You will run out of wheels before you run out of frames and handlebars, which makes the wheels the limiting reactant. Once you run out of wheels, no more complete bicycles can be made.
To determine which reactant is the limiting reactant and which is in excess, it is necessary to calculate the yield of both reactants assuming stoichiometric proportions. The reactant that produces the smaller yield will be identified as the limiting reactant, since it will run out first. Do not assume the reactant in smaller amount will automatically be the limiting reactant.
The answer is 162. Since each bicycle requires 2 wheels, you only have enough wheels to make 162 bicycles (324/2 = 162). You will run out of wheels before you run out of frames and handlebars, which makes the wheels the limiting reactant. Once you run out of wheels, no more complete bicycles can be made.
To determine which reactant is the limiting reactant and which is in excess, it is necessary to calculate the yield of both reactants assuming stoichiometric proportions. The reactant that produces the smaller yield will be identified as the limiting reactant, since it will run out first. Do not assume the reactant in smaller amount will automatically be the limiting reactant.
Example: 30 g of NaCl and 20 g of MgS are mixed together. How many grams of MgCl2 could be produced from the reaction?
Answer: First you need a balanced reaction:
Answer: First you need a balanced reaction:
2 NaCl + MgS --> MgCl2 + Na2S
Then you need to produce the yield of product for the first reactant:
Then you need to produce the yield of product for the second reactant:Then you need to produce the yield of product for the second reactant:
The smaller of the two yields is the correct answer (24.3 g of MgCl2 is the maximum produced). Since the 30 g of NaCl produced a smaller amount of product, the limiting reactant in this case is NaCl.