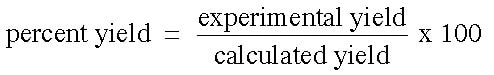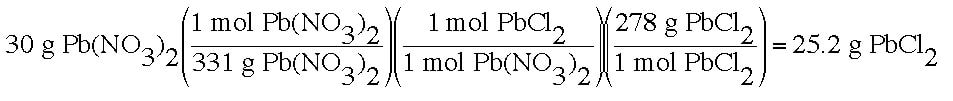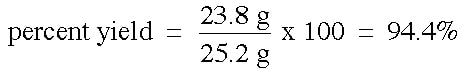Percent Yield
Many times, in performing an experiment, the actual yield (amount you actually produce) will be different from the expected yield (amount you calculate it should produce). In these experiments, a percent yield is helpful. To determine the percent of the calculated yield obtained from the experiment:
Example: In an experiment, we are attempting to make PbCl2 from a mixture of 30 g of Pb(NO3)2 and excess NaCl. Upon completing the experiment, the final measured amount of PbCl2 is 23.8 g. How much lead chloride should have been produced, and what is the percent yield?
Answer:
First, you need a balanced chemical reaction:
First, you need a balanced chemical reaction:
Pb(NO3)2 + 2 NaCl --> PbCl2 + 2 NaNO3
Then, you must calculate the amount that should have been produced by the reaction of 30 g of Pb(NO3)2:
The percent yield is then the percentage produced in the reaction based on the expected yield: