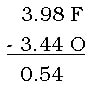Bond polarity
When two atoms attach to one another it is due to the interaction of electrons between the two atoms. These interactions are called bonds. Bonds can form in two different ways:
1) Ionic Bonds – a transfer of electrons from one atom to another causes one atom to become positive, while the other is negative. The opposite ions attract one another and stick together. Ionic bonds occur between metals and nonmetals, or between metals and polyatomic ion groups.
2) Covalent Bonds - bonds that form due to a sharing of a pair of electrons between atoms. In some instances, one atom cannot take away electrons from another, so the two atoms share pairs of electrons, usually one from each atom. This pair of electrons establish the covalent bond, a sharing of electrons due to mutual need of those electrons. Covalent bonds occur between two nonmetal or metalloid elements.
Bonds and Electronegativity
The types of bonds within a compound can be determined by use of an electronegativity chart.
Electronegativity is the ability of an atom to attract electrons within a molecule’s bond. Atoms with high electronegativites have a strong pull on electrons – they really want to gain another electron or two. Atoms with low electronegativity usually do not pull on other atom’s electrons, and in most cases want to get rid of electrons to achieve an octet.
In most cases of ionic bonds, the electronegativity difference between two atoms that is greater than 2.0 will cause an electron to leave one atom and attach to another. In other words, when the electronegativities are far apart, one atom wants electrons a whole lot more than the other, and the bigger electronegative atom takes an electron away. This causes two charged ions, one positive and one negative, which will then attract one another.
There are two types of covalent bonds: polar and nonpolar.
A polar covalent bond exists when atoms share electrons, but the different electronegativities between the two atoms cause a stronger attraction to one atom over the other. The more electronegative atom has a stronger attraction to the electrons, and the electrons of the bond “hang out” more with the more electronegative atom. The more electronegative atom acquires a partial negative charge due to stronger attraction of electrons. The atom with lower electronegativity then has sort of lost it’s complete ownership of the electron that it contributed to the bond, and since it doesn’t have the electron as often as it used to, the atom gets a slight positive charge. Polar covalent bonds occur with an electronegativity difference between 0.5 and 2.0. For example, in the molecule oxygen difluoride, the molecule is polar because when you subtract the electronegativities:
1) Ionic Bonds – a transfer of electrons from one atom to another causes one atom to become positive, while the other is negative. The opposite ions attract one another and stick together. Ionic bonds occur between metals and nonmetals, or between metals and polyatomic ion groups.
2) Covalent Bonds - bonds that form due to a sharing of a pair of electrons between atoms. In some instances, one atom cannot take away electrons from another, so the two atoms share pairs of electrons, usually one from each atom. This pair of electrons establish the covalent bond, a sharing of electrons due to mutual need of those electrons. Covalent bonds occur between two nonmetal or metalloid elements.
Bonds and Electronegativity
The types of bonds within a compound can be determined by use of an electronegativity chart.
Electronegativity is the ability of an atom to attract electrons within a molecule’s bond. Atoms with high electronegativites have a strong pull on electrons – they really want to gain another electron or two. Atoms with low electronegativity usually do not pull on other atom’s electrons, and in most cases want to get rid of electrons to achieve an octet.
In most cases of ionic bonds, the electronegativity difference between two atoms that is greater than 2.0 will cause an electron to leave one atom and attach to another. In other words, when the electronegativities are far apart, one atom wants electrons a whole lot more than the other, and the bigger electronegative atom takes an electron away. This causes two charged ions, one positive and one negative, which will then attract one another.
There are two types of covalent bonds: polar and nonpolar.
A polar covalent bond exists when atoms share electrons, but the different electronegativities between the two atoms cause a stronger attraction to one atom over the other. The more electronegative atom has a stronger attraction to the electrons, and the electrons of the bond “hang out” more with the more electronegative atom. The more electronegative atom acquires a partial negative charge due to stronger attraction of electrons. The atom with lower electronegativity then has sort of lost it’s complete ownership of the electron that it contributed to the bond, and since it doesn’t have the electron as often as it used to, the atom gets a slight positive charge. Polar covalent bonds occur with an electronegativity difference between 0.5 and 2.0. For example, in the molecule oxygen difluoride, the molecule is polar because when you subtract the electronegativities:
the difference is 0.54, which is between 0.5 and 2.0, making it a polar bond. Notice that even though there are 2 Fs in oxygen difluoride, you only subtract one of each atom.
Another way to look at this is through the case of two siblings that get an expensive present for Christmas that they are expected to share. Anyone with brothers or sisters will tell you that sharing is not always equal. In general, the bigger (or more aggressive, uglier, meaner, whinier – you get the picture) child will get to “share”, or play with the present more often that the smaller (or less aggressive) sibling. In this analogy, the electrons in the bond are the present that are shared, and the bigger sibling is the more electronegative atom. The more electronegative atom attracts the electrons to it more powerfully, which allows that atom to become slightly negative. The less electronegative atom becomes slightly positive due to the stronger pull on the electrons from the other atom.
In a nonpolar covalent bond, on the other hand, electrons are shared relatively evenly. This is because the electronegativities of the two atoms that are bonded are similar. The atoms have equal pull on the shared electrons in the bond, so the ends of the bond do not develop a charge. Nonpolar covalent bonds occur when the atoms have an electronegativity difference less than 0.45.
Another way to look at this is through the case of two siblings that get an expensive present for Christmas that they are expected to share. Anyone with brothers or sisters will tell you that sharing is not always equal. In general, the bigger (or more aggressive, uglier, meaner, whinier – you get the picture) child will get to “share”, or play with the present more often that the smaller (or less aggressive) sibling. In this analogy, the electrons in the bond are the present that are shared, and the bigger sibling is the more electronegative atom. The more electronegative atom attracts the electrons to it more powerfully, which allows that atom to become slightly negative. The less electronegative atom becomes slightly positive due to the stronger pull on the electrons from the other atom.
In a nonpolar covalent bond, on the other hand, electrons are shared relatively evenly. This is because the electronegativities of the two atoms that are bonded are similar. The atoms have equal pull on the shared electrons in the bond, so the ends of the bond do not develop a charge. Nonpolar covalent bonds occur when the atoms have an electronegativity difference less than 0.45.