Molecule Polarity
Just like bonds, molecules can be ionic, polar and nonpolar. To decide the polarity of the molecule the following conditions apply:
1) If a molecule has ionic bonds, it is automatically considered an ionic compound.
2) If the molecule has only nonpolar bonds, then the molecule is considered nonpolar.
3) If the molecule has polar bonds, or a combination of polar and nonpolar bonds, it could be either polar or nonpolar. The determining factor will be the shape of the molecule and the location of the bonds. Now that we have studied the shapes, we can put the two concepts together and determine the molecule polarity of a molecule with polar bonds.
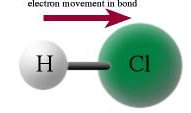
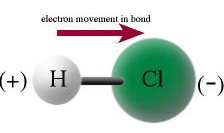
The definition of a polar molecule is a molecule that has a slightly positive end and a slight negative end due to the polar bonds. For example, HCl, the linear molecule with polar bonds has the electrons in the bond pulling toward the chlorine, making the chlorine more negative and the hydrogen more positive.
1) If a molecule has ionic bonds, it is automatically considered an ionic compound.
2) If the molecule has only nonpolar bonds, then the molecule is considered nonpolar.
3) If the molecule has polar bonds, or a combination of polar and nonpolar bonds, it could be either polar or nonpolar. The determining factor will be the shape of the molecule and the location of the bonds. Now that we have studied the shapes, we can put the two concepts together and determine the molecule polarity of a molecule with polar bonds.
The definition of a polar molecule is a molecule that has a slightly positive end and a slight negative end due to the polar bonds. For example, HCl, the linear molecule with polar bonds has the electrons in the bond pulling toward the chlorine, making the chlorine more negative and the hydrogen more positive.
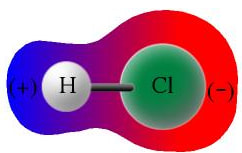
Since, one end of the molecule is positive and the other end is negative, the molecule is polar.
In the gradient picture below, the more blue the area is, the more of a positive attraction it has. The red area is more negative, and attracts negatively.
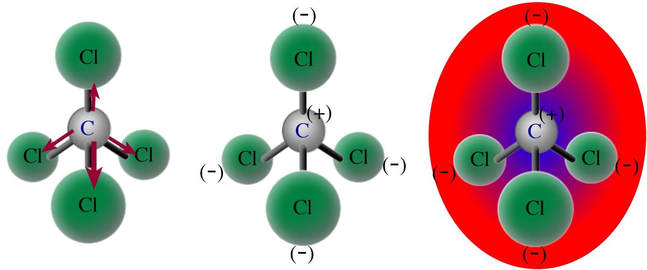
However, not all molecules with polar bonds are polar molecules. In carbon tetrachloride, the molecule is tetrahedral in shape. The chlorine, with a higher electronegativity, pulls the electrons bonded with the carbon toward the chlorines. With all the chlorines having a negative charge, and the carbon with a positive charge, the molecule does not fit under the definition above. In this case, all the ends of the molecule are negative, because the chlorines are all at the outside of the molecule. The positive center is surrounded by negatives. This means, for this molecule, there is not one two ends of the molecule with different charges, so it is a nonpolar molecule.
So, a molecule is polar when two conditions are adhered to:
1) A molecule has polar bonds, and
2) The molecule has one end positive and one end negative, not a situation when one charge is completely surrounded by a different charge.
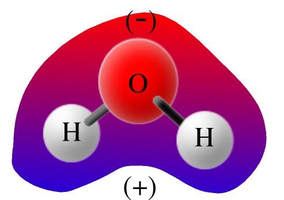
Other examples: Water has positive hydrogens around the negative oxygen, but they are not completely surrounding the negative. Therefore this is a polar molecule. The oxygen is exposed, so one end is positive (the bottom, near the hydrogens) and one end is negative (the oxygen).
1) A molecule has polar bonds, and
2) The molecule has one end positive and one end negative, not a situation when one charge is completely surrounded by a different charge.
Other examples: Water has positive hydrogens around the negative oxygen, but they are not completely surrounding the negative. Therefore this is a polar molecule. The oxygen is exposed, so one end is positive (the bottom, near the hydrogens) and one end is negative (the oxygen).
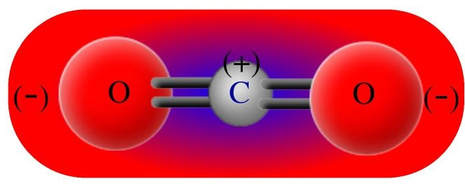
Carbon dioxide has negatives surrounding the positive of a linear molecule. Both ends of the molecule are negative, making the molecule nonpolar.