Energy
Types of energy
Many chemical reactions involve some sort of energy. Energy is the ability to do work or produce heat. There are three general types of energy:
1) Potential energy - energy possessed by objects due to position or arrangement of particles. There are several forms of potential energy
a) electrical - a electrical outlet has the potential to do work. If an appliance is plugged into the outlet, the electricity can perform a function.
b) chemical - a piece of paper can be burned. When the paper is burned, heat and light energy are given off.
c) gravitational - a cart on the top of a cliff can be pushed off the cliff. The cart has the potential to fall.
2) Kinetic energy - energy of motion. Kinetic energy is present in any moving object from a car rolling down a hill to a ball flying through the air.
3) Radiant energy - energy given off by subatomic particles (such as photons, electrons, neutrons, etc.) or chemical reactions. Radiant energy is produced at a source, and travels out in all directions from the source. Examples are heat, light and sound.
Law of Conservation of Energy - Energy can not be created or destroyed, only changed into some other type of energy. Energy conversions can start or end up in potential energy (chemical or position), kinetic energy (mechanical) or radiant energy (thermal, electrical, light, sound). In other words, all energy has to come from somewhere, so a person running is turning chemical potential energy of the food ingested into kinetic energy (of the motion of running) and radiant energy (the heat given off by the person).
Thermochemistry
Thermochemistry is the study of the changes in heat in a chemical reaction. Heat is measured in Joules (J). Heat is different from temperature. Temperature is a measure of the movement of the molecules in a substance, which is measured in degrees.
How is heat different from temperature? Consider this example: On a sunny day, a piece of steel and a piece of Styrofoam are set out in the sun. Assuming that the sun is providing the same amount of energy to both objects, we know that the steel piece will get warm faster, and reach a higher temperature faster than the Styrofoam. Because of the properties of each substance, the metal will increase temperature faster given the same amount of heat energy. Heat is a measure of how much energy is given to an object. Temperature is a measure of how fast the molecules are moving as a result of that heat.
Heat of Reaction
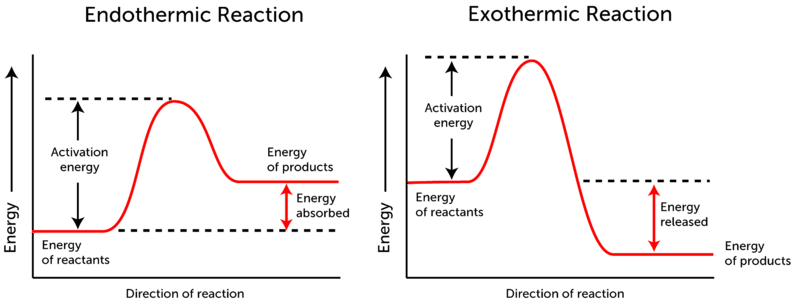
The heat of a reaction depends on the chemical energy of the reactants in comparison to the chemical energy of the products. Most reactions require some energy to get the reaction started. This energy is called the activation energy. The activation energy is the energy necessary to break apart all of the molecules into atoms. Sometimes, the substances have enough energy within them to achieve activation energy, and sometimes more energy is needed. Then, as the bonds reform, the higher amount of energy is used to reform bonds. Typical energy diagrams look like this:
Many chemical reactions involve some sort of energy. Energy is the ability to do work or produce heat. There are three general types of energy:
1) Potential energy - energy possessed by objects due to position or arrangement of particles. There are several forms of potential energy
a) electrical - a electrical outlet has the potential to do work. If an appliance is plugged into the outlet, the electricity can perform a function.
b) chemical - a piece of paper can be burned. When the paper is burned, heat and light energy are given off.
c) gravitational - a cart on the top of a cliff can be pushed off the cliff. The cart has the potential to fall.
2) Kinetic energy - energy of motion. Kinetic energy is present in any moving object from a car rolling down a hill to a ball flying through the air.
3) Radiant energy - energy given off by subatomic particles (such as photons, electrons, neutrons, etc.) or chemical reactions. Radiant energy is produced at a source, and travels out in all directions from the source. Examples are heat, light and sound.
Law of Conservation of Energy - Energy can not be created or destroyed, only changed into some other type of energy. Energy conversions can start or end up in potential energy (chemical or position), kinetic energy (mechanical) or radiant energy (thermal, electrical, light, sound). In other words, all energy has to come from somewhere, so a person running is turning chemical potential energy of the food ingested into kinetic energy (of the motion of running) and radiant energy (the heat given off by the person).
Thermochemistry
Thermochemistry is the study of the changes in heat in a chemical reaction. Heat is measured in Joules (J). Heat is different from temperature. Temperature is a measure of the movement of the molecules in a substance, which is measured in degrees.
How is heat different from temperature? Consider this example: On a sunny day, a piece of steel and a piece of Styrofoam are set out in the sun. Assuming that the sun is providing the same amount of energy to both objects, we know that the steel piece will get warm faster, and reach a higher temperature faster than the Styrofoam. Because of the properties of each substance, the metal will increase temperature faster given the same amount of heat energy. Heat is a measure of how much energy is given to an object. Temperature is a measure of how fast the molecules are moving as a result of that heat.
Heat of Reaction
The heat of a reaction depends on the chemical energy of the reactants in comparison to the chemical energy of the products. Most reactions require some energy to get the reaction started. This energy is called the activation energy. The activation energy is the energy necessary to break apart all of the molecules into atoms. Sometimes, the substances have enough energy within them to achieve activation energy, and sometimes more energy is needed. Then, as the bonds reform, the higher amount of energy is used to reform bonds. Typical energy diagrams look like this:
|
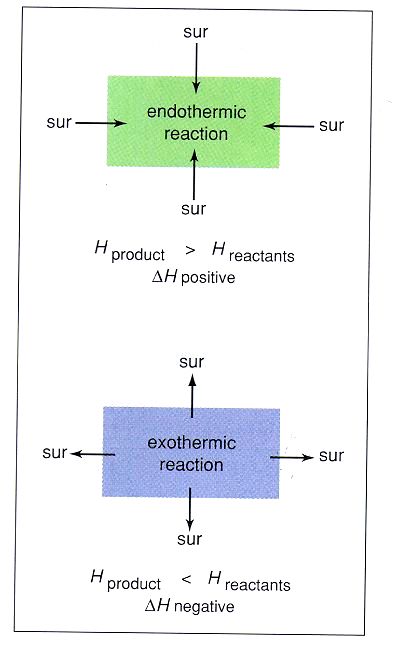
In the endothermic reaction on the left, energy is put into the reaction in order to break all the bonds. That is the activation energy that gets the total energy to the top of the energy "hill". The energy then goes down as bonds are formed. In the endothermic reaction, the energy required to make the products is more than the energy stored in the reactants to begin with. Since there is a net increase in energy (more energy has gone in than released), this is called an endothermic reaction.
The exothermic reaction to the right, still has an activation energy, but after the top of the energy "hill" is achieved, the "fall" in energy to the products ends up below the energy of the reactants. Since, overall, the reactants had more energy in them than the products, the extra energy not used goes out from the reaction. This net decrease in energy (more energy has been lost than gone in) make this reaction exothermic. In general, an endothermic is going to feel cold. This is because the reaction needs energy from outside of it to continue to happen. When you hold a container that contains an endothermic reaction, the heat from your body is sucked into the reaction to form the bonds. This loss of energy from your hand makes it feel cold. An exothermic reaction will feel hot. The excess energy that is released from the reaction will have to go somewhere, so it goes into the surroundings. If your hand is holding a flask that contains an exothermic reaction, the excess energy will go into your hand, and make it feel hot. |