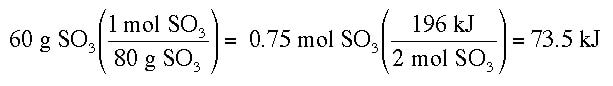Heat of Reaction (Enthalpy)
Enthalpy is a measure of the heat energy absorbed and released in a reaction. Enthalpy is figured as the change in the amount of energy as the reaction continues. The symbol for enthalpy is ΔH. It is based upon the internal potential energy of the reactants (Hreactants) and the internal potential energy of the reactants (Hproducts).
Enthalpy -> ΔH = Hproducts - Hreactants
The enthalpy is either included in the equation:
2 C2H6 + 7 O2 --> 4 CO2 + 6 H2O + 3120 kJ
Or at the end of the reaction:
2 C2H6 + 7 O2 --> 4 CO2 + 6 H2O, ΔH = -3120 kJ
In the case of the above reaction, the reactants contain more potential energy than the products. Therefore the ΔH is negative. A negative enthalpy means the reaction is exothermic because the reactants have lost energy while making the products.
Here are examples of an endothermic reaction. Notice that the when the enthalpy is in the equation, it is on the reactant side, indicating the energy needed to make the reaction occur:
2 SO3 + 196 kJ --> 2 SO2 + O2
At the end of the reaction, the enthalpy is positive, to indicate energy added to the reactants:
2 SO3 --> 2 SO2 + O2, ΔH = 196 kJ
Quiz yourself (answers at bottom):
Endothermic or exothermic?
1) Heat is added to the reaction to make it occur.
2) - ΔH
3) kJ unit on the right side of the equation
4) Heat energy is a product
5) + ΔH
6) kJ unit on the reactants side
Endothermic or exothermic?
1) Heat is added to the reaction to make it occur.
2) - ΔH
3) kJ unit on the right side of the equation
4) Heat energy is a product
5) + ΔH
6) kJ unit on the reactants side
Stoichiometry and Heat
Knowing the enthalpy of a reaction, we can determine the heat given off or absorbed by the given mass of a reactant. The molar coefficients are directly related to the enthalpy, so in an equation if we know how many moles of a substance was used, the moles of that substance are proportional to the enthalpy.
Knowing the enthalpy of a reaction, we can determine the heat given off or absorbed by the given mass of a reactant. The molar coefficients are directly related to the enthalpy, so in an equation if we know how many moles of a substance was used, the moles of that substance are proportional to the enthalpy.
Example: In 2 C2H6 + 7 O2 --> 4 CO2 + 6 H2O + 3120 kJ, 2 moles of C2H6 would give off 3120 kJ of energy. 18 moles of water would give off 9360 kJ of energy (18 is 3 times 6, 9360 is 3 times 3120).
Example 2: Using the equation 2 SO3 + 196 kJ --> 2 SO2 + O2, how much heat would be needed to decompose 60 g of SO3?
Example 2: Using the equation 2 SO3 + 196 kJ --> 2 SO2 + O2, how much heat would be needed to decompose 60 g of SO3?
Answers to quiz:
1) endo
2) exo
3) exo
4) exo
5) endo
6) endo
1) endo
2) exo
3) exo
4) exo
5) endo
6) endo