Petrochemicals and Polymers
Petrochemicals are chemicals that are created from natural gas or oil. Some of these chemicals, when separated from the other substances that are within the mixture of petroleum, can be used immediately in their distilled form (detergents, pesticides, pharmacueticals, etc.) Others require that you take the separated materials, and process it some more to make synthetic substances (plastics, etc.) Plastics include paints, rubber, adhesives, and structural materials.
Builder Molecules
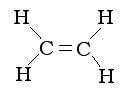
To make these plastics, the double bonds of a monomer, such as ethene, is broken. A monomer is a small molecule with a double bond connecting two carbons in the molecule. A monomer may look like any of these:
Builder Molecules
To make these plastics, the double bonds of a monomer, such as ethene, is broken. A monomer is a small molecule with a double bond connecting two carbons in the molecule. A monomer may look like any of these:
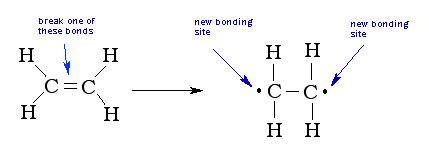
To make a polymer, the double bond of multiple monomers are broken, leaving two open places on each monomer to bond:
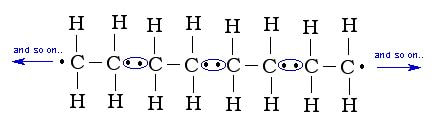
The monomers attach at the new bonding points, and a string of these monomers are chained together to form polymers. These polymers are called addition polymers:
|
The above polymer is called polyethene, as it is made from the monomer ethene. However, since it is a polymerized version of ethene, the name changes and so does the chemical properties. Polyethene is a very common plastic, used as storage containers for many products including milk, detergents and even grocery bags. |
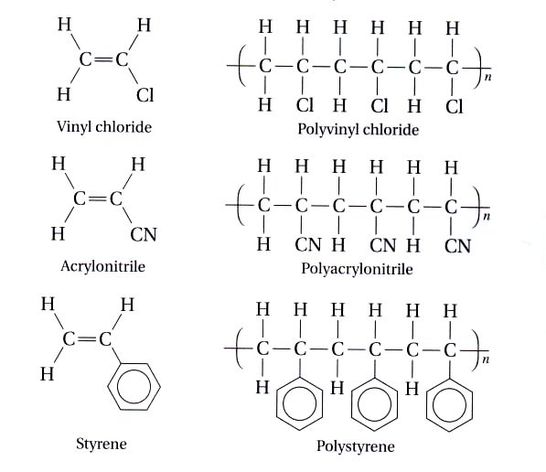
Polymers can be anywhere from a few dozen monomers long, all the way up to millions of monomers long. These polymers are the basis for most plastics that exist today. Plastics are made to different consistency by the length of the polymer and the addition of functional groups. A functional group is an element or compound that replaces one of the hydrogens on the ethane monomer.
Below are examples of the monomers, a look at the polymers, and what they look like in real life.
Below are examples of the monomers, a look at the polymers, and what they look like in real life.