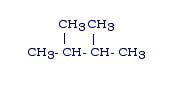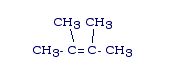Branched Hydrocarbons
Branches off alkanes - Numbering addition
When an alkane has a methyl (-CH3) branch, we must also list where the branch is coming off of the chain. The molecule’s prefix is determined by the length of the longest chain (not including branches), and the number(s) are determined by the places the branch comes off the chain.
Example: 2,3-dimethylbutane
Answer: You need to dissect the name: 2,3 means there are branches coming off the alkane at the 2nd and 3rd carbon. The prefix di- means there are 2 of something. Combined with methyl, that means there are 2 methyl groups coming off the alkane, one off the 2nd and one off the 3rd carbon. The name butane means the main chain of the alkane is 4 carbons long. The structure would look as follows:
Answer: You need to dissect the name: 2,3 means there are branches coming off the alkane at the 2nd and 3rd carbon. The prefix di- means there are 2 of something. Combined with methyl, that means there are 2 methyl groups coming off the alkane, one off the 2nd and one off the 3rd carbon. The name butane means the main chain of the alkane is 4 carbons long. The structure would look as follows:
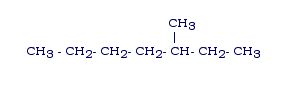
Example 2: Name this alkane:
Answer: 3-methylheptane
Branches off alkenes - Numbering addition
Alkenes are different. When we deal with alkenes with branches, there can be one or two branches:
If there is one branch, the number locating the branch will come before the placement of the double bond.
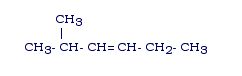
Example: 2-methyl-3-hexene
Branches off alkenes - Numbering addition
Alkenes are different. When we deal with alkenes with branches, there can be one or two branches:
If there is one branch, the number locating the branch will come before the placement of the double bond.
Example: 2-methyl-3-hexene
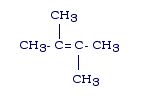
If there are two branches, the branches will come off of the main chain carbons that have the double bond between them. While this is not always the case, as far as we are concerned it will be (It becomes extremely complex).
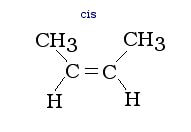
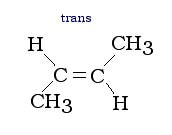
Branches on alkenes of these type can be cis (on the same side), or trans (on opposite sides) to the double bond. The cis and trans isomers have different characteristics from one another, therefore they need to be classified differently. Below are examples of cis and trans branches:
Branches on alkenes of these type can be cis (on the same side), or trans (on opposite sides) to the double bond. The cis and trans isomers have different characteristics from one another, therefore they need to be classified differently. Below are examples of cis and trans branches: