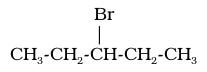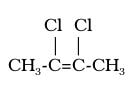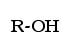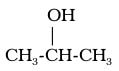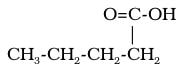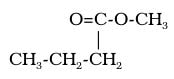Functional Groups
Halogen Functional Groups
A hydrocarbon does not have to be made completely of carbon and hydrogen atoms. If an additional element is included, it is called a functional group. There are many things that can replace a hydrogen on a hydrocarbon. You are already familiar with one functional group: -CH3.
The methyl functional group is really just a branch off of the main carbon chain, but we can consider it a functional group.
Halogens (-F, -Cl, -Br, -I) are very often a functional group. They are named similar to that of the methyl group. In most cases, the addition of a halogen can make the substance more rigid, and less susceptible to reaction.
Halogens (-F, -Cl, -Br, -I) are very often a functional group. They are named similar to that of the methyl group. In most cases, the addition of a halogen can make the substance more rigid, and less susceptible to reaction.
|
Examples: 3-bromopentane
|
cis-2,3-dichloro-2-butene
|
Oxygen Functional Groups
Alcohol, carboxylic acid and ester functional groups can be added to any organic molecule.
Alcohols are present in cleaning products and medicines. The alcohol functional group is an -OH (hydroxyl) group that replaces a hydrogen. Anytime this functional group appears, the hydrocarbon has properties of an alcohol. The general form to designate this group is as follows:
the “R” is an organic chemistry designation. Anytime a chemist sees the “R” symbol, that is supposes to indicate any hydrocarbon, or any carbon hydrogen compound.
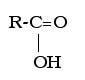
Carboxylic acids are present in natural acids, such as vinegar (acetic acid) and fruits (citric and malonic acids) The carboxylic acid functional group is an -COOH group that replaces a hydrogen, which looks like this, in general form:
Carboxylic acids are present in natural acids, such as vinegar (acetic acid) and fruits (citric and malonic acids) The carboxylic acid functional group is an -COOH group that replaces a hydrogen, which looks like this, in general form:
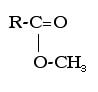
Esters make things smell sweet. They are present in flowers, fruits, candies, etc. The alcohol functional group is an -COOCH3 group that replaces a hydrogen, which looks like this in general form:
Naming for functional groups
As before, these molecules with functional groups are named for the number of carbons. If you start with a hexane core, and replace one hydrogen with one of these functional group, the substance is based off of hexane. Then, you would also need to put a number showing where the functional group is branching off the organic molecule, and then the “e” on the -ane would be replaced by:
As before, these molecules with functional groups are named for the number of carbons. If you start with a hexane core, and replace one hydrogen with one of these functional group, the substance is based off of hexane. Then, you would also need to put a number showing where the functional group is branching off the organic molecule, and then the “e” on the -ane would be replaced by:
a)“-ol” for an alcohol,
b)“-oic acid” for a carboxylic acid, and
c)“1-methyl” prefix and “-oate” suffix for an ester. The methyl part is for the -CH3 group within the ester group…
b)“-oic acid” for a carboxylic acid, and
c)“1-methyl” prefix and “-oate” suffix for an ester. The methyl part is for the -CH3 group within the ester group…