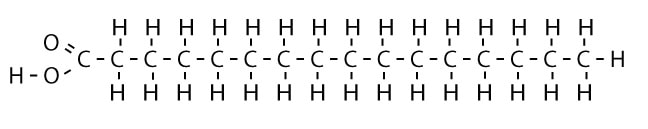Fats
Fats are organic compounds with a long chain of carbon atoms capped with a carboxylic acid group (-COOH) on the end. These molecules, commonly called a fatty acid by organic chemists, are largely insoluble in water and contain a high energy storing capacity. Each gram of fat contains 9 Cal of energy. This has its advantages and disadvantages. Although fats contain a lot of energy for use, they also take more than twice as much energy to burn off a gram. Hence, it is harder (requires more energy) to burn fat than carbohydrates. A simple fatty acid looks like this:
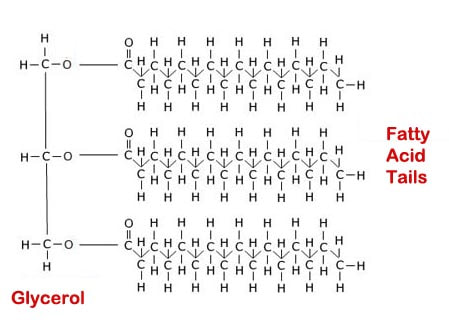
Simple fats are used in cell membranes of plants and animals. When energy is stored as fats in plants and animals, it is stored as a triglyceride, which is 3 fatty acids that are linked together by a glycerol backbone (hence the term triglyceride). These kind of fats are held in the body until needed to make cell membranes or as energy. A typical triglyceride is below. Notice the glycerol connects the three long strings of fatty acids:
Within the fat family, there are two kinds of fats: saturated and unsaturated fats. The saturated fats are saturated hydrocarbons, in which there are no double bonds or triple bonds between carbons. Saturated fats are used in the body to make up the cell membrane of every cell in your body, as well as construction for organs including the brain. However, saturated fats also can absorb into the blood stream and, with LDL cholesterol, can cause blockages in the bloodstream which can lead to heart disease. It is recommended that about 7% of your caloric intake is saturated fat, which figures to be about 160 Cal or about 18 grams of saturated fat per day in a 2000 Cal diet. The picture at the top of the screen, as well as the fat chains in the triglyceride are both examples of saturated fats.
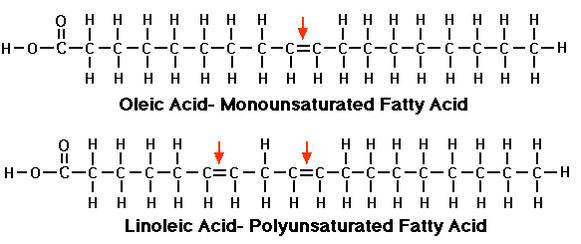
Unsaturated fats are fats that contain at least one double bond between carbon atoms. There are two kinds of unsaturated fats: Monounsaturated (containing one double bond) and Polyunsaturated (containing more than one double bond). Both are used for cell membrane construction, just like saturated, but do not seem to accumulate in the blood stream like saturated fats. Below are an example of mono- and polyunsaturated fats:
Unsaturated fats are fats that contain at least one double bond between carbon atoms. There are two kinds of unsaturated fats: Monounsaturated (containing one double bond) and Polyunsaturated (containing more than one double bond). Both are used for cell membrane construction, just like saturated, but do not seem to accumulate in the blood stream like saturated fats. Below are an example of mono- and polyunsaturated fats:
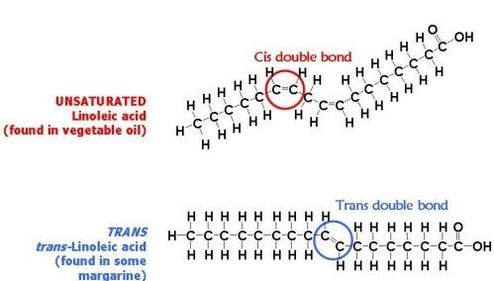
Most dieticians will tell you that if you are going to eat fat, to eat the more liquid unsaturated fats than the solid saturated fats. However, there is a big caveat to that: Trans fats are bad. Unsaturated fats can also be classified as cis or trans fats. Cis fats have the carbons on either side of the double bond on the same side of the molecule (both up or down). In trans fats, the hydrogens are in opposite directions (one up, one down). Cis fats are good. Trans fats, again, are bad. Studies have shown that trans fats can cause many health problems. Most of the issues occur when the trans fat is used to make the cell membrane, which can result in improper regulation of materials moving in and out of the cell. In these cases, if the toxins get into the cell, it can cause replication issues. Below is a picture of a cis and trans fat:
Fats are typically burned in the body after the carbohydrates are used up.