Properties of Gases
|
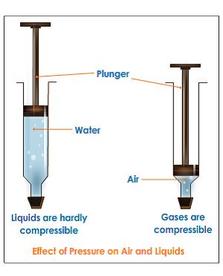
2) Gases are compressible - Squeezing a gas is much easier than squeezing most solids and liquids.
Proof: Placing a gas in a piston, we can reduce the volume of the gas by pressing on the piston. (Figures C). When we place a liquid in the piston, it cannot be compressed. 3) Gases fill containers completely - air is distributed completely throughout a space. Proof: Oxygen is a gas that all people need to survive. Have you ever walked into a part of a room and fallen down dead because there was no oxygen in that part of the room? The answer is no. Gases will fill up their entire container, and be present in all parts of it, whether that container is a room, a building or the atmosphere of the planet. 4) Gases diffuse - gases can move through each other very easily. Proof: Suppose someone were to spill a bottle of perfume in a room. After a certain period of time, that perfume could be smelled throughout the room. This is due to the diffusion of the perfume’s smelly gas molecules through the other gas molecules. |
Figure C
|
5) Gases exert pressure - balloons are given their shape due to the pressure of the gas in the balloon.
Proof: Pressure is a measure of the force of the air molecules pushing on another object. When you try to compress a balloon, the balloon resists the pushing you hand is doing on it. This is due to the gas particles pushing back on the interior of the balloon, which pushes your hands back out.
Kinetic Molecular Theory
The Kinetic Molecular Theory (KMT) is a theory which attempts to explain the behaviors of gases (and solids and liquids, too). There are several assumptions that the KMT makes about gases. They are listed below:
|
1) A gas consists of very small particles, each with a mass. These particles are sub-microscopic, and are considered to be the atoms and molecules of the gas. We know they have a mass because an inflated balloon weighs more than a deflated balloon.
2) The distance between particles of a gas is relatively large. The reason we can compress gases is because there is a lot of space between the particles, and when we compress a gas, we are reducing the space between the particles. 3) Gas particles are in random, constant motion. This is gases can diffuse, because gas particles can move in any direction. 4) Collisions between gases and their container are perfectly elastic. When an object is a gas, the particles bounce off the walls and each other. If a ball were to bounce off a wall, we know that the ball would eventually slow down and come to a rest. Because a balloon stays inflated over a long period of time, we must assume that collisions with the container occur which inflate the balloon. And since the container stays inflated, the particles must keep moving. 5) Gas particles exert no force on one another. The attraction between gas particles must be small because they can bounce off of one another and not get stuck together, The effect is so small we can assume it to be zero. |
Notice that in the animation above, when gas particles collide, sometimes the gas particle will slow down when collisions happen. That energy is transferred into the other particle and the other particle moves faster. Overall, the average speed of the particles remains the same.
|