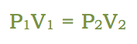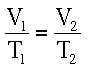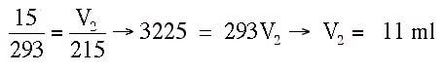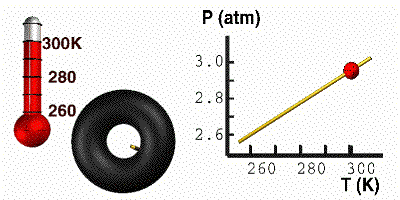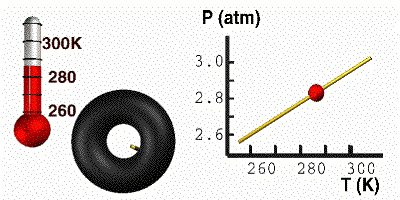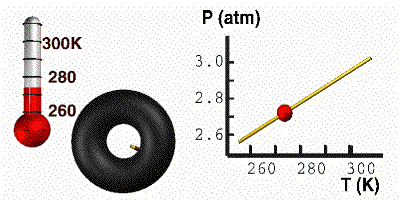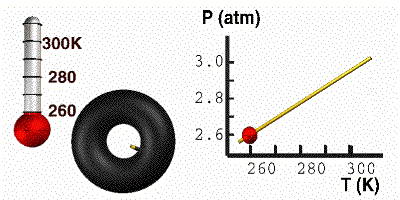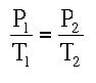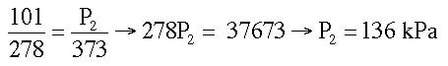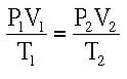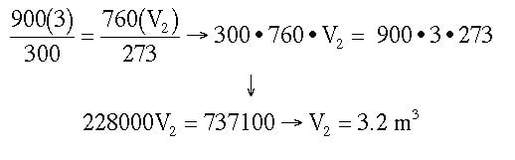Gas Laws
|
Boyle’s Law
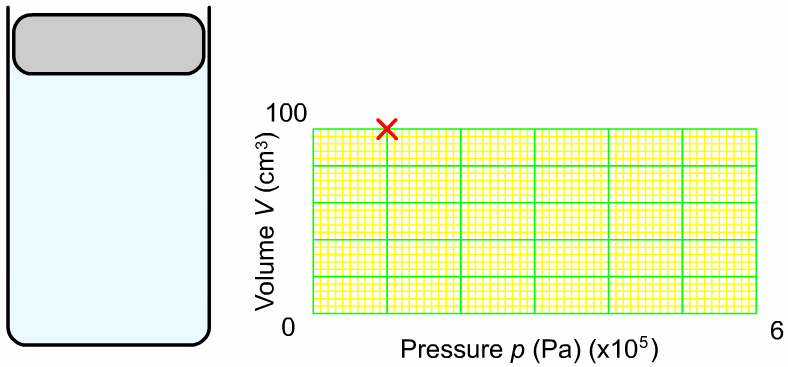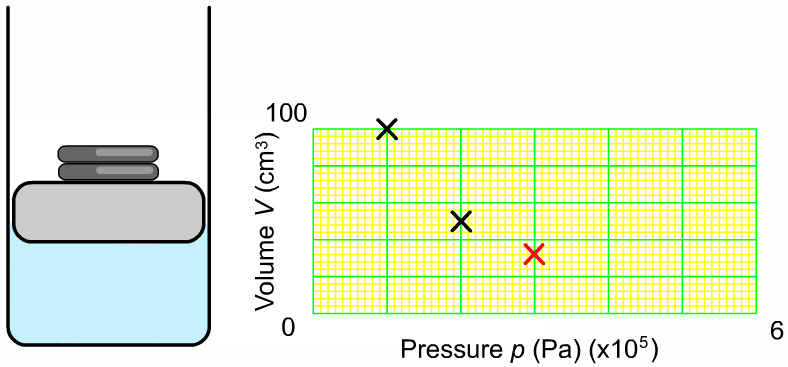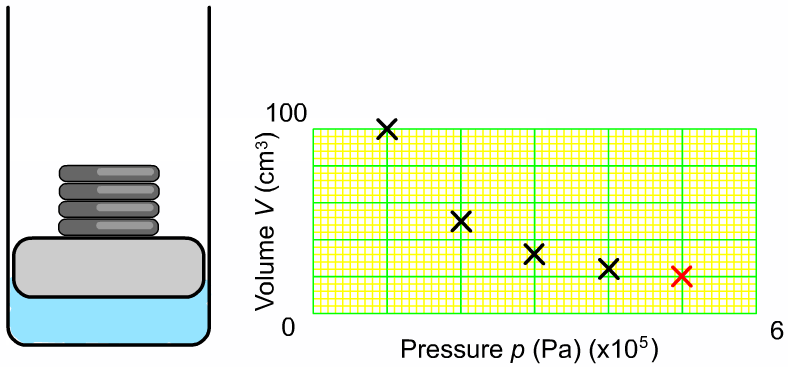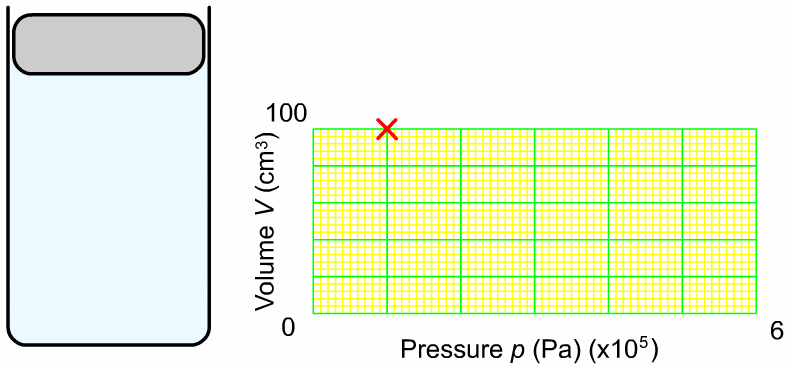
The pressure and volume of a sample of gas at constant temperature are inversely proportional to each other. In other words, as you increase the pressure surrounding a gas, the volume of that gas will go down. Or as you increase the volume of a gas, the pressure that gas provides is lessened. As you can see in the diagram to the right, as you add more weight onto the trapped gas, the volume gets smaller. The greater the pressure applied to the gas, the smaller the volume. The mathematical relationship between pressure and volume is: |
GIF courtesy of iamtechnical.com
|
Where P1 is the starting pressure, V1 is the starting volume, P2 is the final pressure and V2 is the final volume.
Example: A 5 L balloon is placed in a pressure chamber at 745 torr. The pressure is increased to 1800 torr. What is the new volume of the balloon?
Answer: P1 = 745 torr, V1 = 5.0 L, P2 = 1800 torr, V2 = ?
745 (5) = 1800 V2 --> V2 = 2.1 L
745 (5) = 1800 V2 --> V2 = 2.1 L
|
Charles’ Law At constant pressure, the volume of a gas is directly proportional to its absolute temperature. In other words, if the Kelvin temperature of a gas increases, the molecules move faster, they push out on the object containing the gas, and the volume of the gas will also increase. The volume will decrease with a decrease in temperature, because of slower moving molecules. For this equation, temperature must be in Kelvin. Here you have a demonstration of Charles’ Law. The temperature is slowly increased, the volume reacts accordingly. The temperature increases, the particles will move faster, pushing out on the piston more. This causes the volume to expand, as long as the pressure remains constant. The mathematical relationship between volume and temperature is: |
GIF courtesy of iamtechnical.com
|
Where V1 is the starting volume, T1 is the starting temperature, V2 is the final volume and T2 is the final temperature.
Example: An expandable piston holds a volume of 15 ml at 293 K. The temperature of the piston is reduced to 215 K. What is the new volume of the piston?
Answer: V1 = 15 ml, T1 = 293 K, V2 = ? ml, T2 = 215 K
|
Gay-Lussac’s Law
At constant volume, the pressure of a gas is directly proportional to its absolute temperature. In other words, if the Kelvin temperature of a gas increases, the molecules move faster, collide with the container more often, and the pressure of the gas goes up. The pressure will decrease with a decrease in temperature. For this equation, temperature must be in Kelvin. Here you have a demonstration of Gay-Lussac’s Law. On the right, the tire holds a constant volume. As we cool the tire, the pressure responds accordingly. When the temperature goes down, the particles move slower. The particles then bump into the wall less often, causing the pressure to go down. The mathematical relationship between pressure and temperature is: |
GIF courtesy of lovejoyisd.net
|
Where P1 is the starting pressure, T1 is the starting temperature, P2 is the final pressure and T2 is the final temperature.
Example: A steel container holds a gas at 5 OC and 101 kPa. The temperature of the container is increased to 100 OC. What is the new pressure of the container?
Example: A steel container holds a gas at 5 OC and 101 kPa. The temperature of the container is increased to 100 OC. What is the new pressure of the container?
Answer: P1 = 101 kPa, T1 = 278 K, P2 = ? kPa, T2 = 373 K
Combined Gas Law
If none of the values (pressure, volume or temperature) can be kept constant, we can combine the three gas laws into one equation:
If none of the values (pressure, volume or temperature) can be kept constant, we can combine the three gas laws into one equation:
Example: A 3 m3 balloon at 300 K and 900 torr is changed to STP. What is the new volume of the balloon? (Hint: STP stands for Standard Temperature and Pressure, experimental conditions equal to 0 OC and 1 atm)
Answer: P1 = 900 torr, V1 = 3 m3, T1 = 300 K, P2 = 760 torr, V2 = ? m3, T2 = 273 K