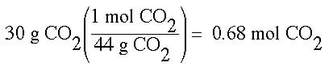Ideal Gas Law
It is possible to combine all the gas laws that we have learned thus far into one gas law. The ideal gas law describes the physical behavior of a gas in terms of the gases’ pressure, volume, moles and temperature. The ideal gas law works for any gas provided it obeys the kinetic molecular theory (KMT) postulates. Deviations from ideal gas occur at very low temperatures and at very high pressures. At low temperatures and high pressures, a gas could turn into a liquid or a solid. If this happens, it is no longer a gas, and will not obey any of the gas laws.
When putting the previously learned gas laws together, we discover the mathematical equation:
PV=nRT
where R stands for the universal gas constant
where R stands for the universal gas constant
The universal gas constant (R) relates a gases’ properties. It is a constant number that relates all of the properties to one another. There are several values for R, which you may use at different times, depending on the units you need. Below is a list of values for R:
|
Values for R:
0.0821 atm⋅L/mol⋅K 8.314 kPa⋅L/mol⋅K 8.314 Pa⋅m3/mol⋅K 62.36 mmHg⋅L/mol⋅K |
When to use:
Pressure is atmospheres, volume is in liters Pressure is kilopascals, volume is in liters Pressure is pascals, volume is in cubic meters Pressure is millimeters of mercury, volume is in liters |
Example: A 30 g sample of CO2 gas has a pressure of 2.5 atm and a temperature of 30 OC. What is the volume of the gas?