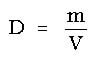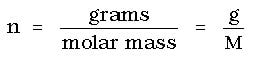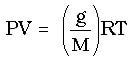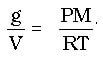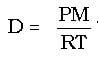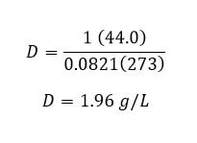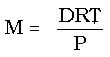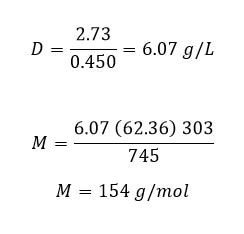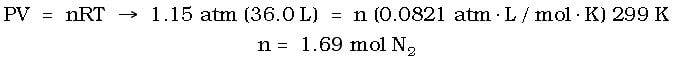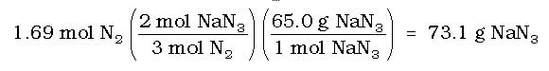Applications of the Ideal Gas Law
The ideal gas law can be used to determine a number of things, including the density of the gas, the gas’s molar mass, and stoichiometric quantities.
Gas Density
Through a rearrangement of the ideal gas law equation, the density of a gas can be derived. Density is a mass per unit volume, or
Gas Density
Through a rearrangement of the ideal gas law equation, the density of a gas can be derived. Density is a mass per unit volume, or
If we replace moles (n) in the ideal gas law with
Then the ideal gas law becomes
This can be rearranged so that g and V are on one side of the equation and the remainder of the quantities of the ideal gas law are on the other as presented here:
Since D = m/V, the ideal gas law has become
In the case of the above reactions, M is the molar mass of the gas in g/mol, Density is determined in the unit of g/L.
Example: What is the density of carbon dioxide at STP?
In order to do this, you have to remember that STP stands for standard temperature and pressure, which is 273 K and 1 atmosphere, respectively. Using the equation above, we need to determine all the components:
Example: What is the density of carbon dioxide at STP?
In order to do this, you have to remember that STP stands for standard temperature and pressure, which is 273 K and 1 atmosphere, respectively. Using the equation above, we need to determine all the components:
Molar Mass of a Gas
Experimentally it is possible to determine the molar mass of a gas, provided you can determine the mass, volume, temperature and pressure at which a gas is collected. From these values, the gas density equation can be rearranged to appear as:
Experimentally it is possible to determine the molar mass of a gas, provided you can determine the mass, volume, temperature and pressure at which a gas is collected. From these values, the gas density equation can be rearranged to appear as:
Example: A gas is collected in a 450 mL flask and is found to have a mass of 2.73 g at 303 K and 745 torr. What is the molar mass of the gas?
Stoichiometry and Ideal Gases
In cases of a chemical reaction in which a gas is either reacted or produced, you may need to use the ideal gas law to determine the moles of gas to complete a stoichiometry problem. As we have talked previously, if the gas is collected at STP, the moles of gas will be proportional to 1 mole = 22.4 L. However, most experiments are not conducted at STP. If a gas is produced or consumed by a chemical reaction that takes place at conditions other than STP, the gas data needs to determine the moles by use of the ideal gas law.
In cases of a chemical reaction in which a gas is either reacted or produced, you may need to use the ideal gas law to determine the moles of gas to complete a stoichiometry problem. As we have talked previously, if the gas is collected at STP, the moles of gas will be proportional to 1 mole = 22.4 L. However, most experiments are not conducted at STP. If a gas is produced or consumed by a chemical reaction that takes place at conditions other than STP, the gas data needs to determine the moles by use of the ideal gas law.
Example: Air bags are inflated by the rapid decomposition of NaN3 as in the reaction:
2 NaN3 (s) --> 2 Na (s) + 3 N(2 (g)
If an air bag must be inflated to a volume of 36.0 L at a pressure of 1.15 atm and a temperature of 26.0 OC, how many grams of NaN3 must decompose?
Answer: First, it is necessary to determine the moles of N2 necessary to fill the bag under the conditions presented above. Through use of the ideal gas law, the moles of N2 will be:
Then, stoichiometry will determine the mass of the NaN3 needed: