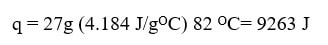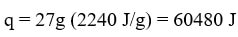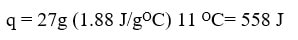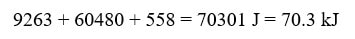Heating Curves (part 2)
Calculating Heat During Phase Change
Example: A 27 g sample of water is heated from 18 OC to 111 OC. How much heat is required to perform this temperature change?
Solution:
Solution:
|
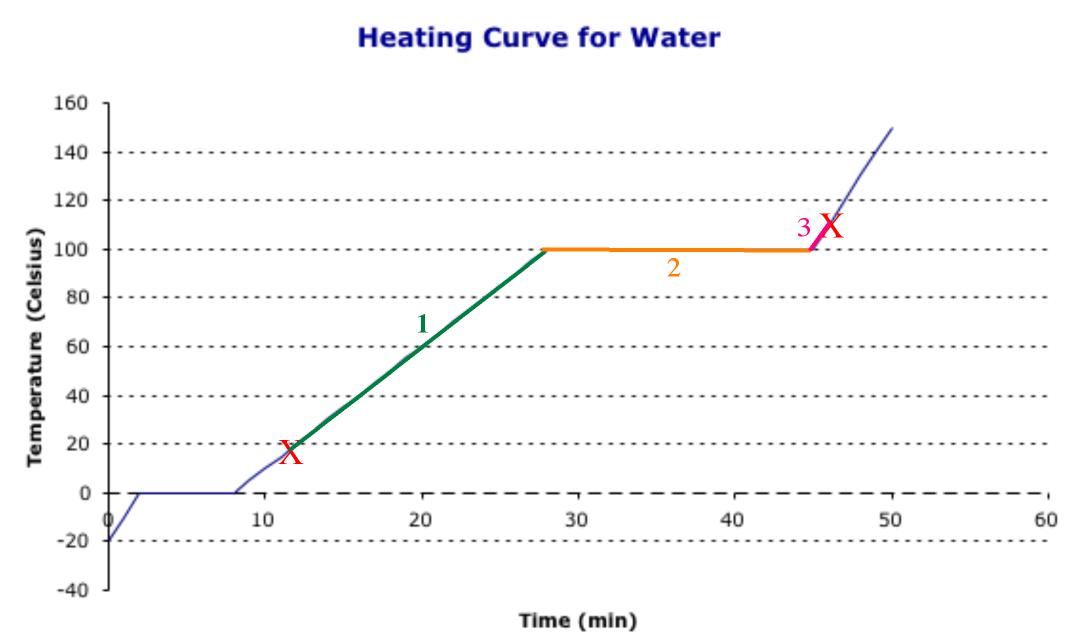
To determine heat needed, it is easiest to think of the calculation as a map. We know what the path looks like, because all heating curves look like the picture to the right.
We can identify the melting and boiling points by the level places in the graph. We know that water freezes at 0 OC and boils at 100 OC. We can mark these places on the graph. Then, we can label our starting point of 18 OC and ending point of 111 OC on the graph. Now, the problem becomes like a map, in which we need to go from start to finish, and determine the heat needed to travel along each of the three roads. The path we follow looks something to the right.
From here, we need to calculate the amount of heat needed on each of the three roads. On road 1, as the temperature changes we can use q=mcΔT: |
On road 2, a phase change is occurring so the temperature does not change. We cannot use q=mcΔT. However, we know the water is boiling, so we can use the heat of vaporization to determine the heat needed to boil the 27 g sample:
On road 3, the temperature of the gaseous water (steam) is increasing so we can use the q=mcΔT equation:
To find the total heat, we then simply add the three values.
For this type of calculation, the maximum number of calculations that must be made before adding would be 5, seeing as how there are only 5 “roads” that we could travel on.