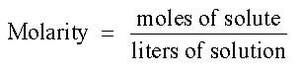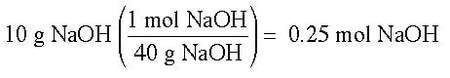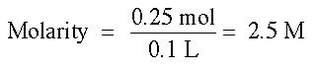MOlarity
Chemists often need to specify precisely how concentrated or dilute a solution is. The concentration is the amount of solute in a given amount of solvent. Earlier this year, we learned some ways of expressing concentration, such as pph, ppm, ppb. These concentrations were based off of masses. Our concentrations need to be more accurate for acids and bases, so we will begin using molarity.
Molarity is the number of moles of solute dissolved in each liter of solution.
The equation for molarity is:
Molarity is the number of moles of solute dissolved in each liter of solution.
The equation for molarity is:
Example: Suppose 10 g of sodium hydroxide (NaOH) were dissolved in 100 ml of water. What would the molarity of solution be?
Answer: To solve for this, we must first determine the moles of solute, which in this case is the NaOH. To determine the moles of NaOH, the following calculation will be done:
Answer: To solve for this, we must first determine the moles of solute, which in this case is the NaOH. To determine the moles of NaOH, the following calculation will be done:
Then, we divide the moles by the liters of solution (100 ml = 0.1 L).
The unit for molarity is a capital M.