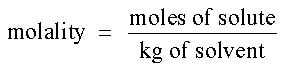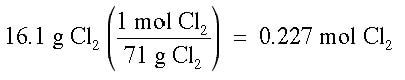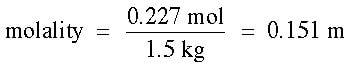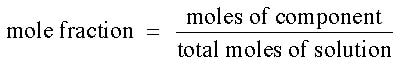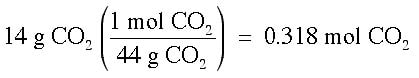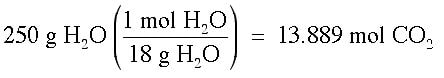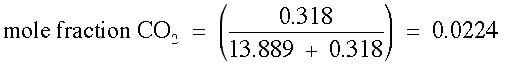More Concentrations
Other more specific ways of expressing concentrations include the following:
Molality is the number of moles of solute dissolved in each kilogram of solvent. The unit for molality is a lower case m. Molality is used when the temperature varies during an experiment. Molarity will change slightly as the temperature changes because volume changes with temperature. The mass and moles in molality do not change with temperature. The equation for molality is:
Molality is the number of moles of solute dissolved in each kilogram of solvent. The unit for molality is a lower case m. Molality is used when the temperature varies during an experiment. Molarity will change slightly as the temperature changes because volume changes with temperature. The mass and moles in molality do not change with temperature. The equation for molality is:
Example: What is the molality of a chlorine solution of 16.1 g of Cl2 dissolved in 1500 g of water?
Answer: It is necessary to change 16.1 g of Cl2 into moles, and 1500 g of water into kg.
Answer: It is necessary to change 16.1 g of Cl2 into moles, and 1500 g of water into kg.
Then, using the molality equation and 1500 g = 1.5 kg:
Mole Fraction is the number of moles of one component divided by the total number of moles in solution. This concentration is similar to pph, but is based on moles instead of grams. There is no unit for mole fraction. The equation for mole fraction is :
Example: What is the mole fraction of CO2 when 14 g of CO2 is dissolved in 250 g of water?
Answer: It is necessary to change each of these grams into moles, then perform the calculation.
Answer: It is necessary to change each of these grams into moles, then perform the calculation.