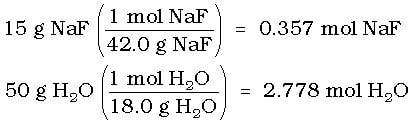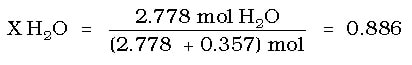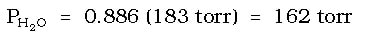Vapor pressure Reduction
|
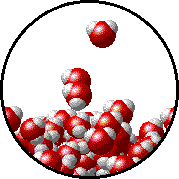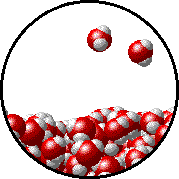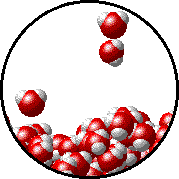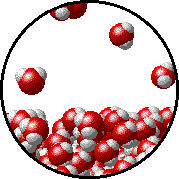
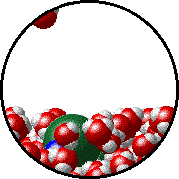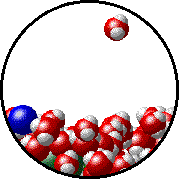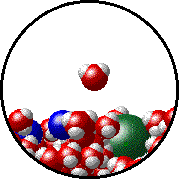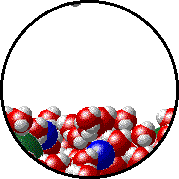
Vapor pressure is due to molecules at the surface of a liquid which break their intermolecular forces and become a gas. As a liquid molecule moves around, it moves faster and slower based on the kinetic molecular theory. On some occasions, a molecule could get bumped by other molecules hard enough that is traveling very quickly. If this molecule is at the surface of the liquid, it could leave the liquid form and become a gas. This process is called evaporation. The newly evaporated gas exerts a pressure on its surroundings. This pressure can be measured. We call it a liquid’s vapor pressure. By adding a nonvolatile (does not evaporate) substance to a liquid, the substance takes up some of the room at the surface of the liquid. This prevents more liquid molecules from being near the surface, kind of creating a dam to prevent molecules from leaving the surface. Some of the liquid molecules can still get through, but not as many as in the pure liquid. Since fewer molecules evaporated create a smaller pressure, the vapor pressure is reduced.
To calculate the new vapor pressure of the liquid, you need two pieces of information. You need what the original vapor pressure of the solvent (liquid). You also need the mole fraction of the solvent. Then, use the following equation: Raoult’s Law: PA = XAPAo
|
Where PA is the new vapor pressure of the gas, XA is the mole fraction of the solvent (that's important - usually we do concentration in terms of the solute) and PAO is the vapor pressure of the pure solvent at the given temperature.
Example: Water vapor pressure of pure water at 55 OC is 183 torr. If 15 g of NaF is added to 50 g of water, what is the new vapor pressure of the solution?
Solution: First we need to find the mole fraction of the water. First calculate the moles of NaF and water:
Then calculate the mole fraction of the water:
Lastly, solve for the new vapor pressure: