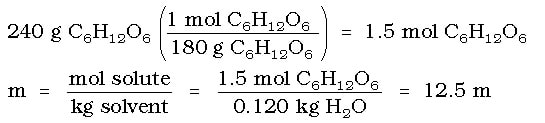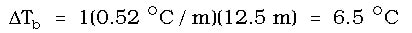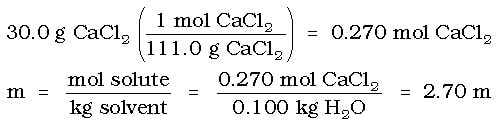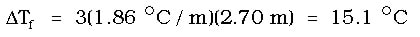Colligative Properties
Boiling Point Elevation
In order for a liquid to boil, the vapor pressure needs to be at the same pressure as the atmospheric pressure above it. As we heat a liquid, the particles begin to move faster. Faster moving molecules have a tendency to leave the surface of the liquid more readily. Therefore, as we heat, the vapor pressure goes up because more molecules of liquid are becoming a gas.
In order for a liquid to boil, the vapor pressure needs to be at the same pressure as the atmospheric pressure above it. As we heat a liquid, the particles begin to move faster. Faster moving molecules have a tendency to leave the surface of the liquid more readily. Therefore, as we heat, the vapor pressure goes up because more molecules of liquid are becoming a gas.
At some temperature of every liquid, the vapor pressure (pressure of the gas trying to escape from the liquid) will become equal to the atmospheric pressure (pressure of gas particles pushing down on the liquid). When this occurs, the liquid will boil. The temperature at which the vapor pressure is equal to atmospheric pressure (1 atm) is the boiling point. For water, the vapor pressure of water is 1 atm when the temperature is 100 OC.
By adding a solute to the liquid, the solution’s vapor pressure is reduced, as we read from above. Since the vapor pressure is now lower at the same temperature, a higher temperature will be required to boil off the liquid. We will need to heat the water above 100 OC in order to get the vapor pressure up to 1 atm.
ΔTb , the difference between the normal boiling point and the new boiling point depends on the molality of the solution:
By adding a solute to the liquid, the solution’s vapor pressure is reduced, as we read from above. Since the vapor pressure is now lower at the same temperature, a higher temperature will be required to boil off the liquid. We will need to heat the water above 100 OC in order to get the vapor pressure up to 1 atm.
ΔTb , the difference between the normal boiling point and the new boiling point depends on the molality of the solution:
ΔTb = iKbm
For this equation, the Kb is the boiling elevation konstant for the solvent. Each solvent has its own Kb. For water, a common solvent, Kb = 0.52 OC/m. "i" represents the level of ionization in the solute. For instance, NaCl will break up into 2 ions when placed in water, so the "i" for a salt water solution would be 2. However, for Ca(NO3)2, the solid would break up into 3 ions (one calcium and 2 nitrates). The "i" would be 3. But, covalent compounds do not break up into ions, so the "i" would be 1 for molecular/covalent compounds.
Example: What is the boiling point of an aqueous glucose (C6H12O6) that has 240 g of glucose dissolved in 120 g of water?
Solution:
Since glucose is a covalent compound, 'i' = 1. Since water is the solvent, the Kb for the solution is 0.52 OC/m. Therefore, we only need to determine the molality to find the temperature difference for the solution. The molality of the solution would be calculated as:
Example: What is the boiling point of an aqueous glucose (C6H12O6) that has 240 g of glucose dissolved in 120 g of water?
Solution:
Since glucose is a covalent compound, 'i' = 1. Since water is the solvent, the Kb for the solution is 0.52 OC/m. Therefore, we only need to determine the molality to find the temperature difference for the solution. The molality of the solution would be calculated as:
Then, we can use the equation above to determine the change in the boiling point:
The change in the boiling point is 6.5 OC, but this does not answer the question. The new boiling point would be this number plus the original boiling point of the pure solvent. Since water boils at 100 OC, we must add 6.5 OC to 100 OC, giving us an answer of 106.5 OC.
Freezing Point Depression
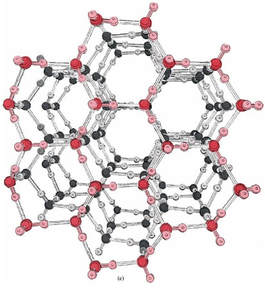
This colligative property is essentially the same as boiling point elevation, except this requires a lower temperature to overcome the molecules of solute getting in the way of intermolecular forces. As molecules freeze, the molecules move slower and slower as the temperature drops. The slow moving molecules set up a lattice that forms a rigid structure. In other words, the puzzle pieces lock together in a pattern. When water freezes, the crystal structure can look like this:
This colligative property is essentially the same as boiling point elevation, except this requires a lower temperature to overcome the molecules of solute getting in the way of intermolecular forces. As molecules freeze, the molecules move slower and slower as the temperature drops. The slow moving molecules set up a lattice that forms a rigid structure. In other words, the puzzle pieces lock together in a pattern. When water freezes, the crystal structure can look like this:
When a large solute molecule is in the solution, the water molecules must form a crystal structure around the solute molecule. In order to do this, the molecules must slow down even more to make a bigger crystal, which requires a lower temperature. The difference between the solvent freezing point and the solution freezing point is ΔTf :
ΔTf = iKfm
where Kf depends on the solvent. For water, the constant Kf = 1.86 OC/m.
Example: What is the freezing point of water when 30.0 g of calcium chloride, CaCl2, is dissolved in 100. g of water?
Solution: Like the above example, we must first determine the molality of the solution:
Example: What is the freezing point of water when 30.0 g of calcium chloride, CaCl2, is dissolved in 100. g of water?
Solution: Like the above example, we must first determine the molality of the solution:
In this case, the CaCl2 is an ionic compound, so it will dissociate into ions. Since we will get 1 ion of calcium and two ions of chloride, the "i" for this solution is 3. Then we must calculate the change in the freezing point:
Again, this is not the answer. Water normally freezes at 0 OC, so the actual freezing point is 15.1 OC below this value, or the solution will freeze at -15.1 OC.