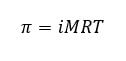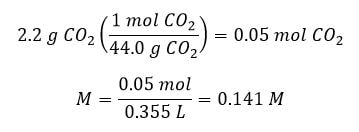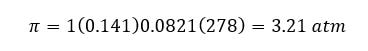osmotic Pressure
|
Osmosis
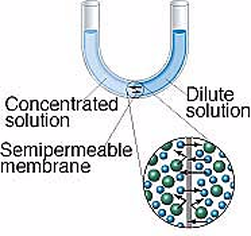
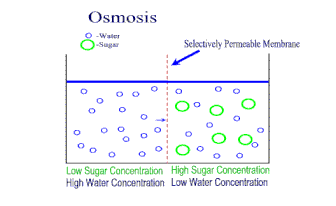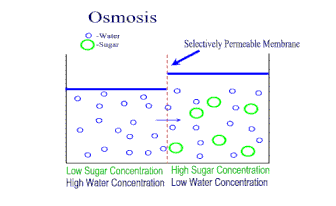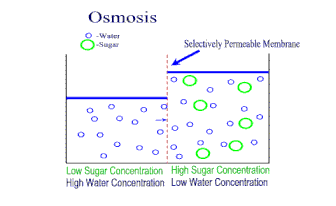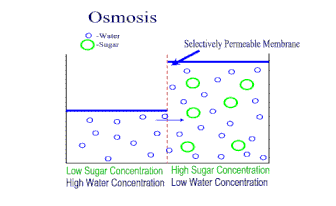
A semipermeable membrane is a material that lets some substances through but not others. It is kind of like a filter, where some things can go through, but not others. However, in this kind of membrane, the "holes" are so small that only very small molecules (like water) are small enough to get through. Dissolved ions, like sodium or chlorine in salt, cannot get through the holes. This makes semipermeable membranes better than filters. Filters allow anything dissolved in the water through. Semipermeable membranes do not. This makes semipermeable membranes better at separation of mixtures. Semipermeable membranes are both natural (every living cell has one - we call it the cell membrane) and we can synthetically make them from petroleum products. There is a natural process in cells called osmosis. When a concentrated solution is separated from a dilute solution by a semipermeable membrane, solvent molecules move from the area of lower concentration to higher concentration. The net movement of solvent is always toward the higher concentration. The molecules of solvent are pulled through the semipermeable membrane to the side of the solution that is more concentrated with solute. The theory is that the water molecules are trying to even out the concentration on both sides of the membrane. The process of osmosis attempts to bring the two concentrations to equilibrium. |
|
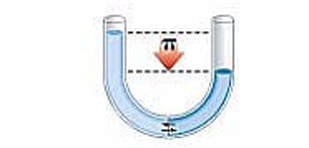
As osmosis continues, the water level on one side of the membrane goes down, and the solution level on the other side goes up. At some point though, the liquid levels of the two solutions becomes so uneven enough that osmosis stops. The weight of the solution on the higher side causes so much pressure on the semipermeable membrane, that the solvent cannot move across the membrane anymore. When this pressure is achieved, osmosis stops. This pressure needed to stop osmosis is called the osmotic pressure.
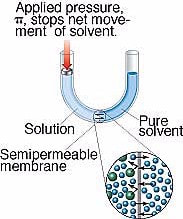
It is possible to apply enough pressure on the solution side in (by use of a plunger) to prevent the flow of solvent through osmosis. This osmotic pressure can be applied to the concentrated solution. If you do this, you must apply enough pressure to stop the osmosis. This osmotic pressure is found to obey the ideal gas law, so that to calculate the osmotic pressure, the following equation is used: Where π is the osmotic pressure, M is the molarity of the solution, R is the universal gas constant and T is the Kelvin temperature
|
Example: A soda solution is found to contain 2.2 grams of carbon dioxide in a 355 mL can of soda solution . If the soda is at 278 K, what would be the osmotic pressure of the solution?
Answer: First we need to find the molarity of the carbon dioxide in the soda:
Then, to find the osmotic pressure, we need the "i". Since carbon dioxide is a molecular compound, the "i" is 1. Substituting the R and temperature into the equation, we get