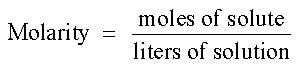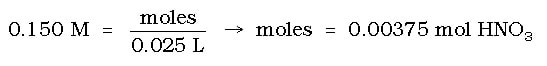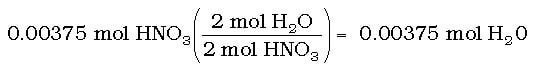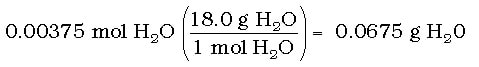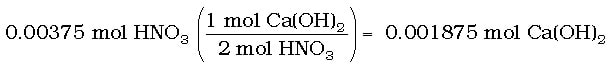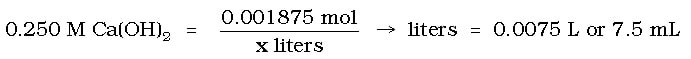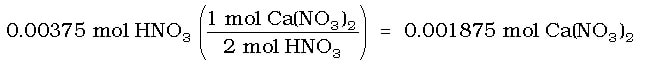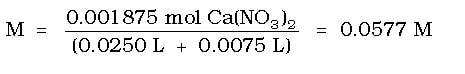Solutions and Stoichiometry
Many times, instead of adding one solid to another in a chemical reaction, to make the reaction go faster we use aqueous solutions. Aqueous solutions contain a certain amount of reactant in a water solution. In most cases, we can determine the amount of solute in solution, in terms of a concentration of solution. If we know the concentration and volume of the materials used. We can use this information to determine moles, and perform the mathematics of a simple stoichiometry problem.
Example:
a) How many grams of H2O (l) are produced when 25.0 mL of 0.150 M HNO3 (aq) are completely neutralized by Ca(OH)2 (aq)?
a) How many grams of H2O (l) are produced when 25.0 mL of 0.150 M HNO3 (aq) are completely neutralized by Ca(OH)2 (aq)?
Solution:
In almost every stoichiometry problem we do, it is first necessary to determine the moles of material we know. From this we can then do a mole-to-mole conversion to find the moles of the stuff we are trying to find, and then ultimately the grams, molecules, atoms, liters or concentration of the new stuff. Our original road map that we have developed now has one more starting and ending place.
In almost every stoichiometry problem we do, it is first necessary to determine the moles of material we know. From this we can then do a mole-to-mole conversion to find the moles of the stuff we are trying to find, and then ultimately the grams, molecules, atoms, liters or concentration of the new stuff. Our original road map that we have developed now has one more starting and ending place.
In this case, we know the concentration and volume of HNO3, so we can determine the moles of HNO3 that needs to be neutralized by using the molarity equation:
Therefore, 25.0 ml of 0.150 M HNO3 can be converted to moles as follows:
Then, we can convert from moles of HNO3 to moles of H2O using the balanced chemical reaction:
2 HNO3 (aq) + Ca(OH)2 (aq) --> Ca(NO3)2 (aq) + 2 H2O (l)
Using these moles of H2O, we can then convert to grams:
b) How many mL of the 0.250 M Ca(OH)2 (aq) were used in this reaction?
Solution: Again, start with the moles of HNO3 from part (a) to find moles of Ca(OH)2:
And then use the molarity equation to find the volume of 0.250 M Ca(OH)2:
c) What salt is produced by this reaction?
The salt produced is the other substance than water, which is Ca(NO3)2 (aq)
d) *What concentration of this salt is made by the neutralization?
Solution: This is just a little tricky at the end. First, we use the moles of HNO3 from part (a) and convert to moles of Ca(NO3)2:
Then, we use the molarity equation to determine the concentration of the salt, but we have to remember that the total volume of the solution is now the combination of the two reactants, the 25.0 mL from the HNO3 and the 7.5 mL from the Ca(OH)2.