Evolution of the Atomic Theory
|
Dalton's Atomic Theory
An atom is defined as the smallest particle of an element that retains the chemical identity of that element. The idea of the atom was originally proposed by a Greek philosopher named Democritus in around 440 BC. Democritus’s model stated that matter consists of invisible particles called atoms and a void (empty space). He stated that atoms are indestructible and unchangeable. Atoms are homogenous, meaning they have no internal structure. His atomic model was solid, and stated all atoms differ in size, shape, mass, position and arrangement, with a void exists between them. This theory had no evidence and was lost for 2200 years. |
In 1803, John Dalton essentially came up with the same theory. Using some experimentation, Dalton theorized that an atom was the smallest form of matter, and that all matter is made from these tiny particles. Dalton assumed that since nothing was smaller than an atom, that atoms would be spherical, and be a solid piece of the element. A picture of what Dalton (and Democritus, for that matter) would believe to be an atom is at right.
Dalton summarized his theory with the following principles.
Dalton summarized his theory with the following principles.
- Each element is composed of extremely small particles called atoms
- All atoms of a given element are identical, but differ from every other element.
- Atoms are neither created nor destroyed in a chemical reaction.
- A given compound always has the same relative number and kinds of atoms.
|
Plum Pudding Model/Electron Discovery
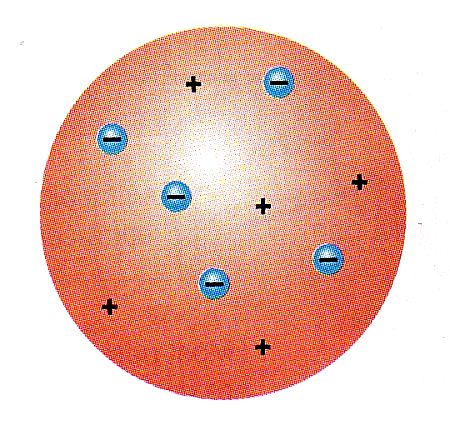
Up until the late 1800’s Dalton's model of the atom held up. Beginning in 1896, J.J. Thomson began working with cathode ray tubes, and discovered that the cathode ray was affected by a magnetic field. With further study, Thomson realized that the cathode ray was actually a stream of negative particles, later called electrons. An example of a cathode ray tube is shown at left. Old-fashioned TV's are modern day versions of cathode ray tubes. Thomson theorized that these electrons had to come from the atom, therefore the atom was comprised of these negative particles (electrons) embedded in a positive (probably gaseous) mass. Therefore, the atom actually contains smaller particles.The model is shown at right. This model is called the plum pudding model, for it reminded Thomson of a dessert called plum pudding. Plum pudding is a bread pudding which has plums that are embedded within the pudding. In contemporary life, we might call it the chocolate chip cookie model. |
Radioactivity
In the 1890’s scientists began working with fluorescence, materials that emit light when hit by radiant EM energy. Two discoveries were very critical to the further development of the atom:
In 1895, W.K. Roentgen discovered X-rays, which react with fluorescence. He therefore assumed that these X-rays were a type of electromagnetic radiation.
In 1896, purely by accident Henri Becquerel discovered that some rocks can expose fluorescence. Becquerel assumed that these rocks naturally give off electromagnetic radiation.
These two discoveries resulted in the development of the theory of radioactivity, that some materials can spontaneously emit radiation. This led to the theory that atoms could be made of something smaller than an atom.
But the discovery of radioactivity produced a bigger question: Where in the atom did the radiation come from?
In the 1890’s scientists began working with fluorescence, materials that emit light when hit by radiant EM energy. Two discoveries were very critical to the further development of the atom:
In 1895, W.K. Roentgen discovered X-rays, which react with fluorescence. He therefore assumed that these X-rays were a type of electromagnetic radiation.
In 1896, purely by accident Henri Becquerel discovered that some rocks can expose fluorescence. Becquerel assumed that these rocks naturally give off electromagnetic radiation.
These two discoveries resulted in the development of the theory of radioactivity, that some materials can spontaneously emit radiation. This led to the theory that atoms could be made of something smaller than an atom.
But the discovery of radioactivity produced a bigger question: Where in the atom did the radiation come from?
|
Discovery of the Nucleus and Proton
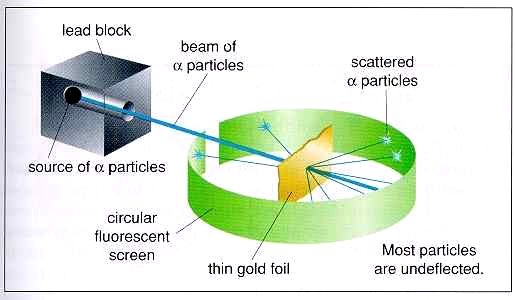
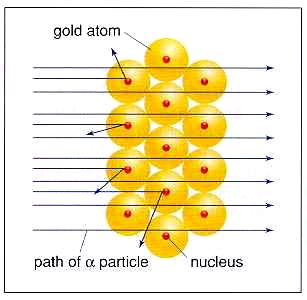
In 1909, Ernest Rutherford performed his Gold-Foil Experiment (shown at right): Alpha particles were fired at a thin layer of gold, with a fluorescent screen behind it. Rutherford theorized that since the gold atoms were mostly a gas with electrons embedded in it (according to Thomson), the alpha particles should pass through them. The majority of the alpha particles passed through, but the foil repelled some of the positive alpha particles. Since alpha particles are positive, there must have been a positive mass in the center of the atom that deflected the alpha particles. Rutherford called this positively charged center the nucleus. Years later, working with other elements in an attempt to make new elements, Rutherford discovered that particles always flew out with a positive charge and the same mass. This particle was called the proton. Since this particle came from the nucleus, he surmised it was part of the nucleus. However, its mass did not equal the mass of the nucleus. Discovery of the Neutron Rutherford proposed that there was another object in the nucleus, without a charge. He called it the neutron, but he had no evidence. In 1932, James Chadwick shot some gamma rays at some beryllium. The radiation that came through the beryllium was not gamma rays, and was not affected by an electric field. He did record the mass of the particle radiation coming from the beryllium, and it had a mass the same as the proton. But it was not charged. This evidence was enough to prove the existence of the neutron. |