Bohr Model of the Atom
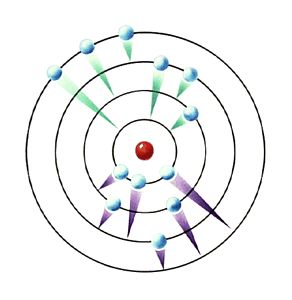
Bohr developed Rutherford’s solar system model of the atom to include energy levels, which we may think of as rings, or orbits, around the nucleus. These energy levels are designated by a quantum number, n. He stated that all electrons of the atom will fill the energy level orbital closest to the nucleus before moving to the next higher orbital. The ground state was the lowest energy level designated by n = 1. These energy levels were simply the orbits around the nucleus, the closest one to the nucleus being n=1, the next n=2 and so forth.
|
Bohr’s theory also provided for a jumping of electrons to an excited state. When this happens, energy is absorbed by the atom, and the electron moves to a higher orbital. This is typically done by electricity. When electricity is run through a sample of an element, the energy provided pushes the electrons out of their normal energy level into a higher energy level. In the diagram to the right, the electrons with the green trails are moving to higher energy levels.
The electron in this higher energy level is unstable, and does not wish to stay there. The electron falls back to its original energy level and as it falls, it gives off quantized energy (E = hf), in the form of electromagnetic radiation. The energy is quantized, which means it is a specific amount of energy. Each different drop gives off different amounts of energy, allowing us to see different bands of light. These drops are represented by the purple trails in the picture to the right. |
When you look at the light given off by hydrogen, it looks purple. But when you look at that light through a spectroscope, you do not see just one color, you see four colors. When these four color are mixed together, it looks purple. But as we can see from the spectroscope diagram below, the four colors are red, green, blue and purple.
|
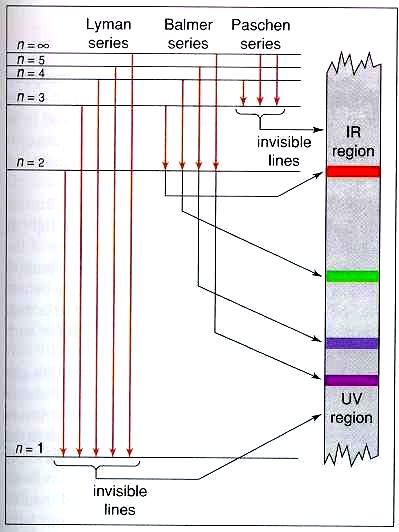
The diagram to the right attempts to explain what transitions in a hydrogen atom cause which of the light bands. Since different colors are seen in the spectra, that means that different drops of the electrons create different colors of light. Each drop represents a different color or type of light that is seen/detected from the hydrogen.
This diagram explains the energy level changes for a hydrogen atom, and which transitions cause which type of light. The horizontal lines on the left represent the different energy levels. The red arrows represent the drops an electron can take. Let’s say an excited electron is in n=3. From there, that electron can drop down to n=2 or n=1. The transition of n=3 to n=2 produces a red band of light. The transition of n=3 to n=1 produces UV light. These two types of light, along with the others can be detected when electricity is run through a sample of hydrogen gas. The point of all this is that Bohr, by proving that different colors of light come from the electrons, showed that the electrons had to be in energy levels to explain the different colors. If there were no different colors, then all the electrons would be in the same level. |