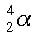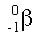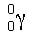Particle Radiation
|
In general, there are three types of radioactive particles. Alpha, beta and gamma radiation can be emitted from any unstable nucleus of an atom. Unstable atoms release this radiation in an attempt to become more stable. Sometimes, multiple decays need to happen for the atom to be come stable. These types of radiation are very different from one another,
An alpha particle is essentially a helium nucleus. Under certain conditions, a large nucleus may give off these particles. An alpha particle is two protons and two neutrons. An alpha particle has a positive charge. An alpha particle in isotopic notation looks like this: |
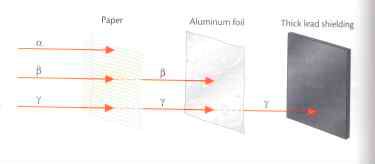
Alpha particles are very large in comparison to beta and gamma. An alpha particle has very low penetrating ability. People can protect themselves against an alpha particle by using a sheet of paper, which will deflect the alpha particle. Human skin is not affected by alpha particles. However, internal organs that have little to no protection can be harmed by alpha particles. Alpha particles, although easily blocked, can cause a good deal of damage to internal organs because of their size. Therefore, alpha particles are considered ionizing radiation.
A beta particle is essentially a free electron. When a beta particle is released, the electron comes flying out of the nucleus. A beta particle has a negative charge. A beta particle in isotopic notation looks like this:
A beta particle is essentially a free electron. When a beta particle is released, the electron comes flying out of the nucleus. A beta particle has a negative charge. A beta particle in isotopic notation looks like this:
Beta particles, which are much smaller than alpha particles, have more penetrating ability. A piece of aluminum foil is required to deflect a beta particle. Beta particles are ionizing radiation, and can penetrate the skin and cause damage to skin and nerve cells.
Gamma radiation is produced from the release of pure energy from a nucleus. A gamma ray has no mass and no charge, it is simply a stream of high frequency energy. A gamma particle in isotopic notation looks like this:
Gamma radiation is produced from the release of pure energy from a nucleus. A gamma ray has no mass and no charge, it is simply a stream of high frequency energy. A gamma particle in isotopic notation looks like this:
Gamma rays have no mass, which makes them more difficult to stop. Protection against gamma radiation requires several inches of lead to shield against the radiation. Gamma radiation can travel through every component of the human body, and the radiation when absorbed can cause genetic mutations in living cells.
A diagram of the three particles penetrating ability is shown below:
A diagram of the three particles penetrating ability is shown below: