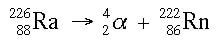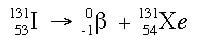Radioactive Decay (or Nuclear Decay)
Radioactive decay is a natural phenomenon of some large atom isotopes. Some isotopes of atoms have unstable nuclei. Unstable nuclei over time desire to become stable. These isotopes accomplish this by ejecting alpha, beta or gamma radiation from the nucleus.
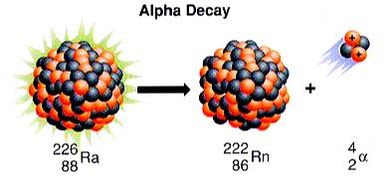
Alpha decay is characterized by the release of an alpha particle from an unstable nucleus. The nucleus releases this alpha particle in order to achieve stability. When the nucleus releases the alpha particle, the number of protons decreases by two, which means that the element changes. Below is an example of an alpha decay.
Alpha decay is characterized by the release of an alpha particle from an unstable nucleus. The nucleus releases this alpha particle in order to achieve stability. When the nucleus releases the alpha particle, the number of protons decreases by two, which means that the element changes. Below is an example of an alpha decay.
As you can see, the release of the alpha particle changes the nucleus from radium (Ra) to radon (Rn). Below is another way of looking at the same reaction:
|
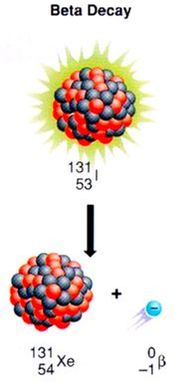
Beta decay is characterized by the release of a beta particle from an unstable nucleus. The nucleus releases this beta particle in order to achieve stability. When the nucleus releases the beta particle, the number of protons increases by one, which means that the element changes. Below is an example of a beta decay.
The atomic number changes in the nucleus because beta decay occurs from the decomposition of a neutron. In beta decay, the neutron breaks up into a proton and an electron:
Because electrons do not belong in the nucleus, the electron gets ejected from the nucleus and escapes the atom, becoming a free electron, or a beta particle. To the right is another way looking at the same reaction: Gamma decay is the loss of pure energy from the unstable isotope. In a gamma decay, the element does not change. In this nuclear reaction, the nucleus simply rearranges itself into a tighter form (kind of like the settling in a box of cereal). When this rearranging occurs, it takes less energy to hold the atom together, and the extra energy is released in the form of a gamma ray. An example of a gamma decay is shown below: As you can see, the element stays the same.
|