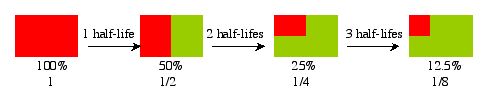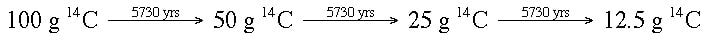Half-Life
Half-life is the amount of time it takes for one half of the original radioactive material to decay into a new substance. Radioactive materials will alpha, beta or gamma decay over time. Since it takes a period of time for something to decay, the half-life measures the amount of time needed for half of the radioactive material to change into something more stable.
All materials have radioactive isotopes within them. Even living things have radioactive elements in them. When these objects are first created, or when a plant or animal eats something, it acquires some of these radioactive isotopes. These radioactive isotopes are replenished in a living being as long as it is alive. When a living being dies, the number of radioactive isotopes does not replenish because the being stops eating. Once the object dies, the number of radioactive isotopes will decrease over time as the isotope decays. Knowing that the amount of radioactive isotopes will decrease over time, the age of an object can be determined.
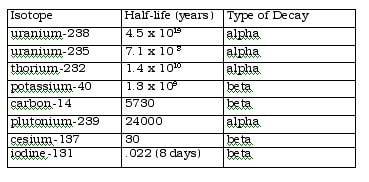
Examples of radioactive isotopes and their half-life:
All materials have radioactive isotopes within them. Even living things have radioactive elements in them. When these objects are first created, or when a plant or animal eats something, it acquires some of these radioactive isotopes. These radioactive isotopes are replenished in a living being as long as it is alive. When a living being dies, the number of radioactive isotopes does not replenish because the being stops eating. Once the object dies, the number of radioactive isotopes will decrease over time as the isotope decays. Knowing that the amount of radioactive isotopes will decrease over time, the age of an object can be determined.
Examples of radioactive isotopes and their half-life:
Suppose you had 16 g of iodine -131. After one half-life (8 days), the amount of iodine-131 would be down to 8 g. After another 8 days (one half-life) the amount of iodine-131 would be down to half of what it was, or down to 4 g. After another 8 days (one half-life) the mass of iodine-131 would be 2 g. It keeps cutting the amount in half every half-life. Theoretically, the amount of radioactive isotope would never get down to zero.
Another way to look at it would be:
Another way to look at it would be:
In this example, the red material represents all of the radioactive isotope. After 1 half life, the amount of radioactive material is cut in half (50 % red) and the new material formed (the green) is 50 % too. However, after another half life, the amount of red material gets cut in half again, bringing the red material down to 25%. The new material is now 75% of the mass. The process continues on, each time cutting the red in half. The red material, however, will never get down to zero (theoretically).
Example of use of half-life.
A fossil is found in the ground. Based on the fossil size, the creature should have had 100 g of carbon-14 when it was living. It is determined that the fossil has 12.5 g of carbon-14 currently. If the half-life of carbon-14 is 5730 years, then how old is the fossil?
If the fossil originally contained 100 g of carbon-14, after one half-life (5730 years) the fossil would contain 50 g. After another half-life (5730 years), the amount of carbon-14 would decrease by 1/2 again and be down to 25 g. After another half-life (5730 years), the amount of carbon-14 would decrease by 1/2 again and be down to 12.5 g. The fossil then would be 3 half-lifes x 5730 yrs = 17,190 years old
Another way to look at it would be:
Example of use of half-life.
A fossil is found in the ground. Based on the fossil size, the creature should have had 100 g of carbon-14 when it was living. It is determined that the fossil has 12.5 g of carbon-14 currently. If the half-life of carbon-14 is 5730 years, then how old is the fossil?
If the fossil originally contained 100 g of carbon-14, after one half-life (5730 years) the fossil would contain 50 g. After another half-life (5730 years), the amount of carbon-14 would decrease by 1/2 again and be down to 25 g. After another half-life (5730 years), the amount of carbon-14 would decrease by 1/2 again and be down to 12.5 g. The fossil then would be 3 half-lifes x 5730 yrs = 17,190 years old
Another way to look at it would be: