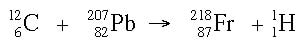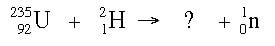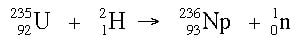Transmutation (Artificial Radioactivity)
Transmutation is the process of converting one element into another using bombardment with high energy particles. Transmutation, or artificial radioactivity, is when you hit a nucleus with a projectile. If the projectile hits the nucleus at a great enough rate, the projectile and the nucleus will fuse together and make a larger nucleus. In most cases, the nucleus will eject a by-product particle.
Below is an example of a transmutation reaction.
Below is an example of a transmutation reaction.
The target nucleus is the isotope which is bombarded (in the example above, the lead-207)
The projectile is the particle fired at the nucleus (carbon-12)
The product is the new nucleus produced by the reaction (francium-218)
The ejected particle is the light nucleus or particle emitted in the reaction (hydrogen-1)
It is possible to predict the product of a transmutation reaction if you know what the target nucleus and the projectile are (it is also helpful to know if there are any ejected particles).
Example: What is the product nucleus when a uranium-235 nucleus is hit with a hydrogen-2 nucleus, and a neutron is ejected from the target?
Answer: So far we know this much:
The projectile is the particle fired at the nucleus (carbon-12)
The product is the new nucleus produced by the reaction (francium-218)
The ejected particle is the light nucleus or particle emitted in the reaction (hydrogen-1)
It is possible to predict the product of a transmutation reaction if you know what the target nucleus and the projectile are (it is also helpful to know if there are any ejected particles).
Example: What is the product nucleus when a uranium-235 nucleus is hit with a hydrogen-2 nucleus, and a neutron is ejected from the target?
Answer: So far we know this much:
By the law of conservation of matter, the product side of the reaction should add up to the reactant side in terms of the atomic number. If the reactant side has a mass of 237, then the product side needs to have a mass of 237 as well. By subtraction (237 - 1), the product nucleus should be 236. A similar calculation can be done for the atomic number, and the answer becomes: