Atoms and Molecules
Pure Substances, or elements and compounds, can be differentiated into two categories, depending on their makeup.
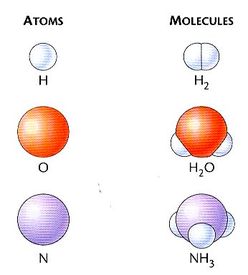
1) Atoms are the building block of matter. Atoms are the smallest part of an element that still retains the physical properties of that element. If you took a piect of aluminum foil and cut it in half millions of times, eventually you would have to cut two atoms apart. This one atom of aluminum has the exact same physical and chemical properties as the group of aluminum atoms in the aluminum foil.
2) Molecules are two or more atoms chemically bonded to one another. In molecules, you need to have at least two atoms that are bonded together, not just mixed together.
Models of atoms and molecules:
1) Atoms are the building block of matter. Atoms are the smallest part of an element that still retains the physical properties of that element. If you took a piect of aluminum foil and cut it in half millions of times, eventually you would have to cut two atoms apart. This one atom of aluminum has the exact same physical and chemical properties as the group of aluminum atoms in the aluminum foil.
2) Molecules are two or more atoms chemically bonded to one another. In molecules, you need to have at least two atoms that are bonded together, not just mixed together.
Models of atoms and molecules:
Elements can appear as atoms or as molecules. Aluminum can exist as an atom (however, we wouldn’t be able to see it). Hydrogen cannot exist by itself. Hydrogen has to be combined with another hydrogen into a molecule in order to be stable.
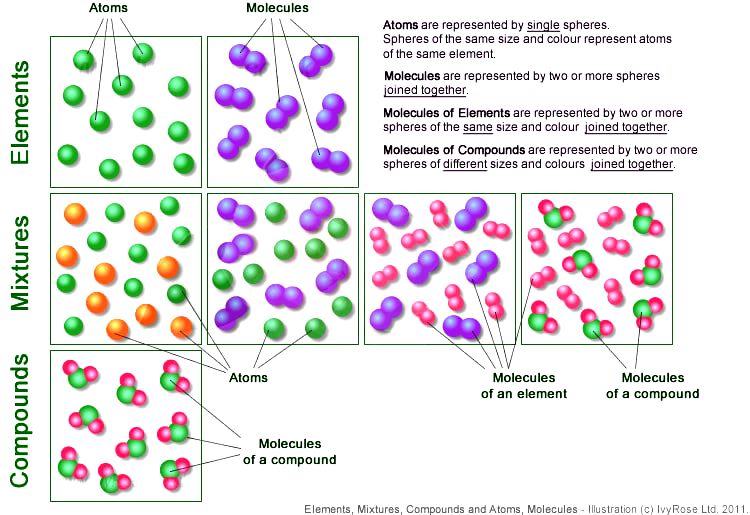
Compounds are always molecules, because to make a compound, you have to have 2 different elements bonded together.
The below pictures are depictions of various mixtures of compounds, elements, atoms and molecules.
Compounds are always molecules, because to make a compound, you have to have 2 different elements bonded together.
The below pictures are depictions of various mixtures of compounds, elements, atoms and molecules.