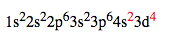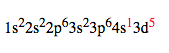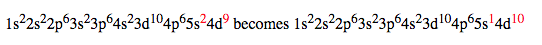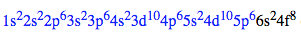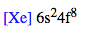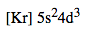Applications of Configurations
Exceptions to the Aufbau Principle
When the Aufbau Principle is applied to all elements, certain elements do not agree experimentally with Aufbau. In other words, sometimes the lower energy orbital does not have to be filled to put an electron in a higher orbital. Due to subtle electron interactions, a new configuration is formed which allows the atom to be more stable. The exception is applied when the configuration ends in any of the following ways: d4, d9, f6, or f13. For example, chromium is one of those elements. Chromium, with 24 electrons, would normally have the following configuration:
However, to make the atom more stable, one of the electrons from the 4s orbital jumps into the 3d orbital, changing the configuration to:
Another example: Silver with 47 electrons
For the f-orbital changes, again one electron is taken from the s-orbital and moved up to the f-orbital to change it to f7 or f14.
Shortcuts for Configurations
When writing configurations for large atoms, the pattern becomes repetitive. It is allowed (in order to save time) to abbreviate the configuration by listing the noble gas prior to the element [in parenthesis], then finishing off the configuration from there. The configuration for gadolinium (64 electrons) can be written:
When writing configurations for large atoms, the pattern becomes repetitive. It is allowed (in order to save time) to abbreviate the configuration by listing the noble gas prior to the element [in parenthesis], then finishing off the configuration from there. The configuration for gadolinium (64 electrons) can be written:
The xenon atom has a configuration of 1s22s22p63s23p64s23d104p65s24d105p6 always, so to the reader, the [Xe] means that the atom starts with a xenon core (or 1s22s22p63s23p64s23d104p65s24d105p6) and continues from there.
Ex. Niobium (41 electrons) -
It would be helpful for this purpose to know what the configurations for the noble gases are. This picture should help: