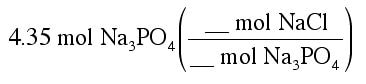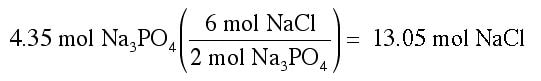Stoichiometry
Stoichiometry
Stoichiometr is from the Greek language, in which stoicheion meaning element and metron meaning measure.
Stoichiometry is the study of quantitive (measurable) relationships that exist in chemical formulas and chemical reactions. In other words, we are attempting to determine the amount of material made in a chemical reaction based on the knowledge of just one component.
Suppose we had the following balanced equation:
Stoichiometr is from the Greek language, in which stoicheion meaning element and metron meaning measure.
Stoichiometry is the study of quantitive (measurable) relationships that exist in chemical formulas and chemical reactions. In other words, we are attempting to determine the amount of material made in a chemical reaction based on the knowledge of just one component.
Suppose we had the following balanced equation:
N2H4 + 2 H2O2 --> N2 + 4 H2O
From this balanced equation, we know that when we mix 1 molecule of N2H4 with 2 molecules of H2O2, then 1 molecule of N2 and 4 molecules of H2O will be produced. The coefficients in front of the materials tell us the quantities of each needed to complete the reaction.
Now, we could multiply every coefficient by the same number, say 20, and still have a balanced equation, or:
Now, we could multiply every coefficient by the same number, say 20, and still have a balanced equation, or:
20 N2H4 + 40 H2O2 --> 20 N2 + 80 H2O
As long as all coefficients are multiplied by the same number, then the equation will remain balanced:
N2H4 + 2 H2O2 --> N2 + 4 H2O
|
N2H4
1
3000 6.02 x 1023 1 mole 7 moles |
+
|
2 H2O2
2
6000 2(6.02 x 1023) 2 mole 14 moles |
-->
|
N2
1
3000 6.02 x 1023 1 mole 7 moles |
+
|
4 H2O
4
12000 4(6.02 x 1023) 4 mole 28 moles |
As you can see, the two rows in blue have identical numbers. The upper row is molecules, the lower row is moles, but the numbers are the same. Therefore, the coefficients not only tell us the molecule ratio in the reaction, it also tells us the mole ratio.
Mole to Mole Stoichiometry
Knowing these facts, it is now possible to predict the amount of chemicals used or produced in a reaction from knowing just one amount of any one of the materials.
Let’s start with a different reaction:
Mole to Mole Stoichiometry
Knowing these facts, it is now possible to predict the amount of chemicals used or produced in a reaction from knowing just one amount of any one of the materials.
Let’s start with a different reaction:
3 BaCl2 + 2 Na3PO4 ---> 6 NaCl + Ba3(PO4)2
Suppose we know that we are going to mix 4.35 mol of Na3PO4, and we want to know how many moles of NaCl are made. We can use the fact that the equation tells us the ratio of moles to set-up the following conversion:
Click to set custom HTML
The numbers that go in the conversion come from the balanced chemical reaction. Because a 6 comes before NaCl, we put a 6 in the top of the conversion factor. And, since a 2 comes before Na3PO4, we put a 2 in the bottom of the conversion factor:
This answer should make sense. We know that the sodium chloride moles is always 3 times more than the sodium phosphate because the mole ratio of sodium chloride to sodium phosphate from the chemical reaction are 6:2, which is also 3:1.