Petroleum
|
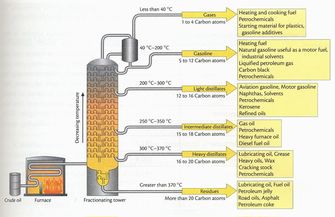
Petroleum, or crude oil, is a mixture of molecular substances that results from the decaying of plants and animals. Over thousands of years, as massive amounts of dead plant and animal materials accumulates and decays, the earth processes will decompose it into several organic molecules. These molecules range from very small to very large hydrocarbons that are typically called alkanes. This mixture of alkanes cannot be used as it is pumped out of the ground. Rather, it needs to separated into parts in order to be used.
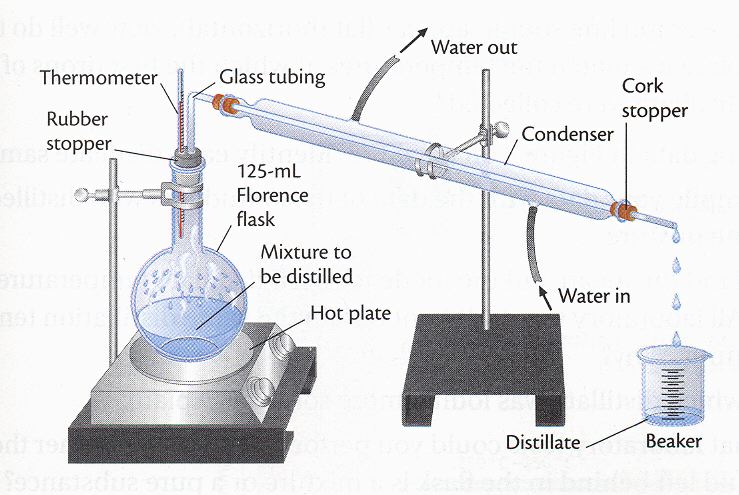
Distillation is a form of separation. When two materials are mixed, and cannot be separated by means like filtration, another common method is distillation. In distillation, one of the components of the mixture is boiled in a flask, while the temperature is not warm enough to boil the other components. The gas travels up through the tube, and travels down the condensation tube. The cooler condensation tube cools the gas back down, and turns it back into a liquid. The liquid then drips into the collection flask. The two liquids are now separated. (See figure). Fractional Distillation When crude oil (petroleum) is heated to 400 oC, many of the components of the mixture turn into a gas. The gas is then pumped into a column, diagrammed at right. As the column extends into the air, the temperature inside the column goes down. In other words, the farther you get from the fire, the lower the cooler the temperature. As the vapor rises in the column, the temperature cools, and when the gas reaches the temperature in the column that it turns back into a liquid, there are little collection bowls that collect the condensed liquid. The higher the gases travels up in the column, the lower its boiling point.
|
|
Intermolecular Forces
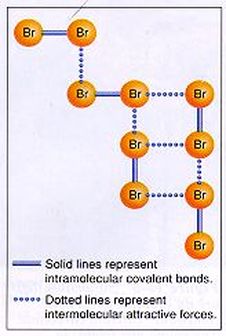
Why do alkanes have different boiling points? Boiling and condensing are caused by intermolecular forces. Intermolecular forces hold solids and liquids together. Intermolecular forces can be thought of as “glue”. In solids and liquids, there is a “glue” that holds the water molecules together. This “glue” does not allow any of the water molecules escape. However, when the temperature goes up, the molecules move faster, and it makes it more difficult for the “glue” to hold onto the molecules. When the boiling point of water is reached, the molecules are moving so fast, the “glue” cannot hold the molecules together, and a gas is formed. The higher the boiling point, the stronger the intermolecular forces. Therefore, substances with low boiling points (easy to boil) have weak intermolecular forces and substances with high boiling points (difficult to boil) have strong intermolecular forces. In the diagram at left, there are two types of forces: intramolecular forces (the bonds) and intermolecular forces (the “glue”). The intermolecular forces are the forces of attraction between neighboring molecules. (inter - outside). The intramolecular forces, or the bonds, are the stronger forces. A bond is always going to be stronger than the intermolecular forces that hold solids and liquids together. In order to break an intermolecular force, heat needs to be added to boil the substance. In order to break a bond, much more aggressive means are necessary. |