Food Energy Storage
Food Energy
All food energy originates from sunlight. From the energy of light from the Sun, plants use sunlight to undergo an endothermic reaction that converts smaller molecules into larger molecules. This process of photosynthesis is often depicted as:
All food energy originates from sunlight. From the energy of light from the Sun, plants use sunlight to undergo an endothermic reaction that converts smaller molecules into larger molecules. This process of photosynthesis is often depicted as:
6 CO2 (g) + 6 H2O (l) + 2800 kJ --> C6H12O6 (s) + 6 O2 (g)
The glucose molecules (C6H12O6) are made as the plants take in carbon dioxide from the air and water from the ground. The radiant energy from the sun gets transformed into chemical potential energy stored within the bonds of the glucose molecules. Only plants can perform this function. Animals cannot cause photosynthesis to occur in their bodies. These stored energy molecules are where animals get their energy. Animals eat the glucose molecules (among other molecules with potential energy) and use the stored energy in the bonds.
Animals have the ability to use food energy immediately. Through a complex version of a combustion reaction, carbohydrates (like glucose) along with proteins and fats can be burned to release the stored energy: The reaction for the burning of a carbohydrate like glucose is:
C6H12O6 (s) + 6 O2 (g) --> 6 CO2 (g) + 6 H2O (l) + 2800 kJ
Notice that this reaction is the exact reverse reaction of photosynthesis. Also, notice that this reaction releases 670 Calories per mole of glucose. In other words, if you eat 1 mole of glucose (180 g), you will need to burn 670 Calories in order to “work it off”.
Other carbohydrates, fats and proteins have similar combustion reactions that release similar amounts of energy. The energy released by this reaction can be used for any activity in the cells, which allows your organs to function and your muscles to move. When this energy is released, it can be used immediately by absorption into the cells, or the energy can also be stored for later use.
Energy Storage
In cells, the amount of energy needed to perform the various functions is much less than what is provided for by the “burning” of glucose. Plus, you are not eating all of the time, so the energy from the glucose needs to be stored for times when energy is not coming in. If our bodies had no way of storing energy, we would have to eat all the time just in order to breathe and pump our heart. So, most of the potential energy is burned, released to the cells where the cells transform the energy into another potential energy source that is housed in the cell.
Other carbohydrates, fats and proteins have similar combustion reactions that release similar amounts of energy. The energy released by this reaction can be used for any activity in the cells, which allows your organs to function and your muscles to move. When this energy is released, it can be used immediately by absorption into the cells, or the energy can also be stored for later use.
Energy Storage
In cells, the amount of energy needed to perform the various functions is much less than what is provided for by the “burning” of glucose. Plus, you are not eating all of the time, so the energy from the glucose needs to be stored for times when energy is not coming in. If our bodies had no way of storing energy, we would have to eat all the time just in order to breathe and pump our heart. So, most of the potential energy is burned, released to the cells where the cells transform the energy into another potential energy source that is housed in the cell.
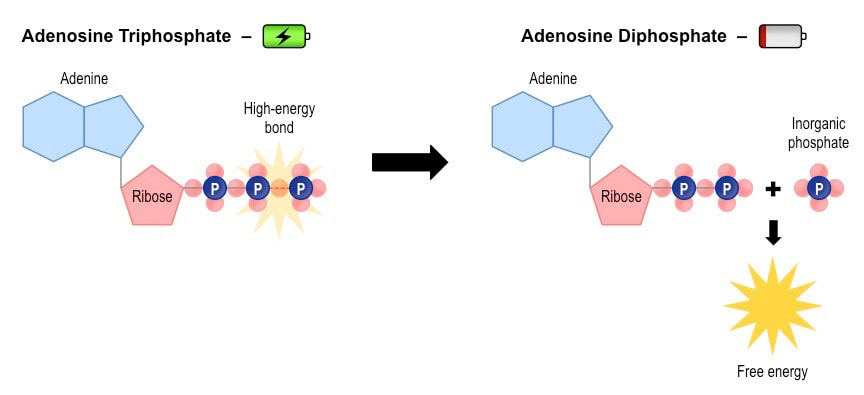
In cells, the amount of energy needed to perform the various functions is much less than what is provided for by the “burning” of glucose. Within the cells are biomolecules called adenosine diphosphate (ADP) and adenosine triphosphate (ATP) which store the energy in small amounts to be used by the cell later. The only difference between ADP and ATP is the addition of a phosphate (PO4-3) ion. di- means two and tri- means three, so the addition of a third phosphate to the adenosine is the difference in these molecules. It takes a lot of energy to add a third phosphate to adenosine, and that energy is then stored in the bond between the second and third phosphate. A diagram for this is shown below:
This additional phosphate bond is a high energy bond that can be used later. As the cell is required to replicate, move or any other function it performs, the cell can break the third phosphate bond of ATP to become ADP. The energy is then absorbed by the organelles in the cell to perform whatever function is called for. In this case, the reaction reverses, loses the phosphate ion which reacts with water, and then the reaction can occur again when more energy is absorbed.