Carbohydrates
|
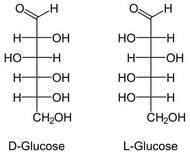
Originally, chemists analyzed the structure of sugars and found that the formula was always Cx(H2O)x. They determined this molecular structure similar to how we could do this in a laboratory, including dehydration and subtraction of mass. Because it looked like water was attached to carbon, the name carbohydrate was used. However, this was just an empirical formula and the chemists had no idea what the structure was like.
|
|
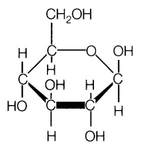
In the end, through processes using powerful machinery like a gas chromatograph it was later determined that the structures for simple sugars looked like the diagram to the right. Obviously, there is no water in the structure. For simplification, carbohydrates are ringed structures of carbons where each carbon has a hydroxyl functional group (-OH). This is an oversimplification, but for our purposes it will be sufficient.
|
Types of sugars
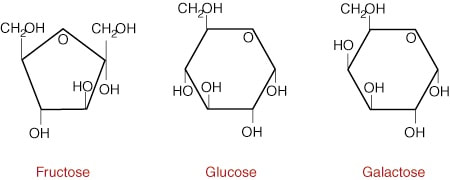
Monosaccharides are simple sugars, like glucose, that are about 5 to 6 carbons long (or ringed). Examples include glucose (blood sugar), fructose (fruit sugars) and galactose (simple milk sugar). The structures of these sugars are very similar, with each structure having only a slightly different arrangement (some are even isomers of one another). The picture above, and those just below are monosaccharides.
Monosaccharides are simple sugars, like glucose, that are about 5 to 6 carbons long (or ringed). Examples include glucose (blood sugar), fructose (fruit sugars) and galactose (simple milk sugar). The structures of these sugars are very similar, with each structure having only a slightly different arrangement (some are even isomers of one another). The picture above, and those just below are monosaccharides.
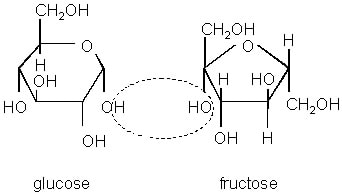
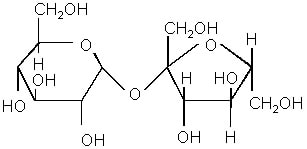
Disaccharides are a combination of two monosaccharides, where water is released in a condensation reaction. A condensation reaction is another way to make a polymer, similar to an addition reaction. However, in this reaction, water is a byproduct of the reaction. A common condensation reaction is depicted below:
These are identifiable by their structure, in which it appears as if the ringed structure repeats itself. Examples include sucrose (table sugar), lactose (milk sugar) and maltose (malt sugar).
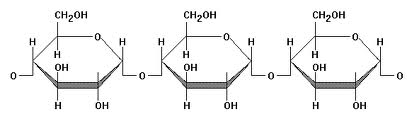
Polysaccharides is a polymer version of a sugar. When condensation reactions are repeated over and over again, and many rings of monosaccharides are added onto one another to produce long chains, a polymer is formed. In the sugars, this polymer is called a polysaccharide. Examples include starch (plant stored sugars), cellulose (plant cell wall sugars) and glycogen (animal stored sugars). A generic polysaccharide is shown below. Keep in mind that a polysaccharide (like a polymer) must be 100’s of individual units long to be considered a polymer.
Polysaccharides is a polymer version of a sugar. When condensation reactions are repeated over and over again, and many rings of monosaccharides are added onto one another to produce long chains, a polymer is formed. In the sugars, this polymer is called a polysaccharide. Examples include starch (plant stored sugars), cellulose (plant cell wall sugars) and glycogen (animal stored sugars). A generic polysaccharide is shown below. Keep in mind that a polysaccharide (like a polymer) must be 100’s of individual units long to be considered a polymer.
Energy from Carbs
In most human diets, the majority of the energy that humans use are due to the burning of sugars and starches. Most nutritionists recommend 60% of your daily intake of Calories should come from carbohydrates. Each gram of a carbohydrate provides about 4 Cal of food energy. The carbohydrate is the easiest for the body to break down. The breakdown of starches starts in the mouth when saliva provides an enzyme to break down the starch into simple sugars. Once broken down, the body can easily absorb the sugars into the blood stream for transport to the cells. This process is quite efficient in the human body, and carbohydrates are broken down first.
In most human diets, the majority of the energy that humans use are due to the burning of sugars and starches. Most nutritionists recommend 60% of your daily intake of Calories should come from carbohydrates. Each gram of a carbohydrate provides about 4 Cal of food energy. The carbohydrate is the easiest for the body to break down. The breakdown of starches starts in the mouth when saliva provides an enzyme to break down the starch into simple sugars. Once broken down, the body can easily absorb the sugars into the blood stream for transport to the cells. This process is quite efficient in the human body, and carbohydrates are broken down first.