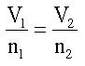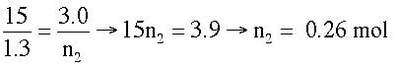Additional Gas Laws
Avogadro’s Principle
Equal volumes of gases under the same conditions have equal number of molecules. In other words, if you have two balloons of equal volume, under the same pressure and temperature, then those two balloons will have an equal number of molecules (or moles) in them. This may sounds like it makes complete sense, but the Principle says that the gas in the two balloons do not have to be the same gas…one could be He and the other be CO2. If the two balloons has equal volumes, then there would be the same number of moles of He as CO2.
Earlier this year, you were told that 1 mole = 22.4 L at STP. STP are experimental conditions, where the experiment was performed at 0 OC and 1 atm of pressure. When we used 22.4 L = 1 mole before, we assumed that all problems and experiments were at STP. Now, we know that not everything is at STP, so we need to keep that in mind as we do problems.
The mathematical relationship between volume and moles is:
Earlier this year, you were told that 1 mole = 22.4 L at STP. STP are experimental conditions, where the experiment was performed at 0 OC and 1 atm of pressure. When we used 22.4 L = 1 mole before, we assumed that all problems and experiments were at STP. Now, we know that not everything is at STP, so we need to keep that in mind as we do problems.
The mathematical relationship between volume and moles is:
Where V1 is the starting volume, n1 is the starting moles, V2 is the final volume and n2 is the final moles.
Example: A balloon has 1.3 moles of gas in and has a volume of 15 L. Gas is released from the balloon until the volume is 3.0 L. How many moles are now in the balloon?
Answer: V1 = 15 L, n1 = 1.3 mol, V2 = 3.0 L, n2 = ?
|
Dalton’s Law of Partial Pressures
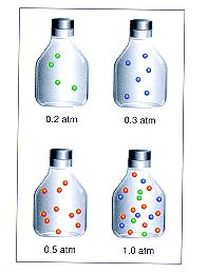
The total pressure in a gas mixture is the sum of the partial pressures of the individual components. Partial pressures are the pressures due to each gas in the mixture. Look at the picture at right. The blue gas in the bottle has a pressure of 0.2 atm. The green gas in the bottle has a pressure of 0.3 atm. The red gas in the bottle has a pressure of 0.5 atm. Each of these individual partial pressures will combine when all three gases are placed in the bottle at the same time. The total pressure of the gas mixture is the 0.2 + 0.3 + 0.5 = 1.0 atm. The mathematical relationship is: Ptotal = P1 + P2+ P3 + ...
The … is to show you can add as many gases together as you need, from only 2 to 20 or more.
|
Example: A mixture of 70% N2 and 30% O2 has a total pressure of 210 psi. What are the partial pressures of each gas?
Answer: N2: 0.70 (210) = 147 psi N2 ; O2: 0.30 (210) = 63 psi O2
Answer: N2: 0.70 (210) = 147 psi N2 ; O2: 0.30 (210) = 63 psi O2