Heating Curves (part 1)
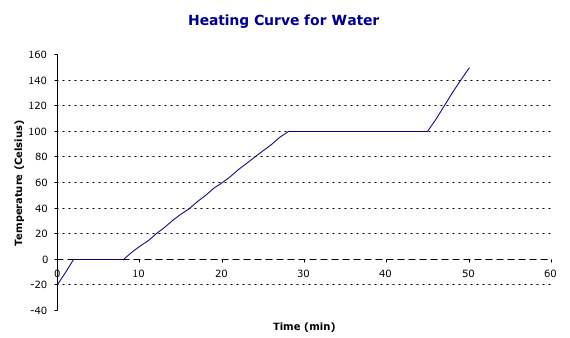
As a solid material is heated from a solid to a liquid to a gas with constant heat, there are two abnormalities to the curve. We believe that heat and temperature influence one another. As we add heat something, the material’s temperature should increase in a linear fashion. However, as a solid material is heated from a solid to a liquid to a gas with constant heat, there are two abnormalities to the curve. The two abnormalities occur at the melting point and at the boiling point of the materials. At these points, the temperature remains virtually constant until the material completely changes state.
|
So, as heat is added to a piece of ice, the temperature of the ice will increase to 0 OC. At this point, the temperature will remain constant until all the ice melts into water. Then, once the water has all melted, the temperature begins to rise again. Now, the temperature will continue to increase until the water reaches 100 OC, the boiling temperature. At this point, the temperature will remain constant until all the water has boiled into steam. After the water has boiled completely into steam, the steam's temperature will increase.
As you can also see from the graph, the slope of the three slanted lines are not all the same. This is not a manipulation of the data. For all substances, as you use constant heat, the solid liquid and gas phases all heat at a different rate, or have a different specific heat depending on the state of matter. |
So, as heat is added to a piece of ice, the temperature of the ice will increase to 0 OC. At this point, the temperature will remain constant until all the ice melts into water. Then, once the water has all melted, the temperature begins to rise again. Now, the temperature will continue to increase until the water reaches 100 OC, the boiling temperature. At this point, the temperature will remain constant until all the water has boiled into steam. After the water has boiled completely into steam, the steam’s temperature will increase.
As you can also see from the graph, the slope of the three slanted lines are not all the same. This is not a manipulation of the data. For all substances, as you use constant heat, the solid liquid and gas phases all heat at a different rate, or have a different specific heat depending on the state of matter.
For example, with water the specific heats are
|
solid ice
liquid water gaseous steam |
2.09 J/gOC 4.184 J/gOC 1.88 J/gOC |
So, what that means is you have to use a different specific heat depending on whether you are dealing with the solid, liquid or gas version of the material. For instance, say you had 30 grams of ice at -5 OC, and you cooled it to -23 OC. How much heat would need to be removed?
Well, as before, it is necessary to use the equation q = mcΔT, so first we determine each part:
Well, as before, it is necessary to use the equation q = mcΔT, so first we determine each part:
|
q = ?
m = 30 g c (for ice) = 2.09 J/gOC ΔT = -23 -(-5) = -18 OC |
q = 30 (2.09) -18 = -1128 J (or 1128 J removed)
|
Heat of Vaporization & Fusion
The temperature of the substance does not increase until all the solid has melted. It also does not increase until all the liquid is boiled off. The amount of heat needed to melt a substance is called its heat of fusion. The hear of fusion of water is Hf = 334 J/g. In other words, it takes 334 J of energy to melt one gram of solid ice into liquid water. Subsequently, to freeze one gram of water, 334 J of energy needs to be removed from the water.
The heat of vaporization of water is Hv = 2240 J/g for water. It takes 2240 J to boil one gram of water into steam.
The heat of vaporization of water is Hv = 2240 J/g for water. It takes 2240 J to boil one gram of water into steam.
Just like specific heat, every substance has a different heat of fusion and heat of vaporization value. The values listed above are for water only. To find heats of fusion and vaporization for other substances, a resource would be needed.
So, by the same token, we can figure out how much heat it takes to freeze, melt, boil or condense a material.
Example: How much energy is needed to boil 25 grams of water?
|
q = ?
m = 25 g Hv = 2240 J/g |
q = 25(2240) = 56000 J
|